Colorectal cancer remains one of the leading causes of cancer-related morbidity and mortality worldwide. Advances in chemotherapy have introduced novel drug combinations that improve patient survival and quality of life. Among these, the fixed-dose combination of Trifluridine and Tipiracil, marketed as Lonsurf®, has gained significant clinical importance. Trifluridine, a thymidine analog, exerts its antineoplastic activity by incorporating into DNA and inhibiting DNA synthesis (1, 2). However, its rapid degradation by thymidine phosphorylase limits its therapeutic potential. Tipiracil, a thymidine phosphorylase inhibitor, enhances the bioavailability of Trifluridine by preventing its catabolism, thereby ensuring sustained plasma concentrations and improved efficacy (3, 4). Given the therapeutic relevance of this combination, accurate and reliable analytical methods are essential for quality control, stability testing, and regulatory compliance. High-performance liquid chromatography (HPLC) has emerged as the method of choice for pharmaceutical analysis due to its sensitivity, reproducibility, and ability to separate complex mixtures. While individual methods for Trifluridine and Tipiracil have been reported, simultaneous estimation using a single RP-HPLC method offers efficiency, reduced analysis time, and cost-effectiveness in routine laboratory practice (5, 6). The present study focuses on the development and validation of a simple, precise, and robust RP-HPLC method for the simultaneous estimation of Trifluridine and Tipiracil in bulk and tablet dosage form. The method was validated in accordance with ICH Q2(R1) guidelines, covering parameters such as linearity, accuracy, precision, specificity, robustness, and system suitability (7, 8). This work aims to provide a reliable analytical tool for routine quality control and pharmaceutical research involving this clinically significant drug combination.
MATERIALS AND METHODS
2.1 Drugs and Chemicals
Trifluridine and Tipiracil reference standards were procured from Sigma-Aldrich Chemicals Pvt. Ltd., Bengaluru, India, ensuring pharmaceutical-grade purity for analytical studies. The marketed tablet formulation containing Trifluridine (20 mg) and Tipiracil (8.19 mg) was obtained from Taiho Oncology’s Lonsurf® tablets, sourced through an authorized distributor in India. Acetonitrile (HPLC grade) was supplied by Merck Life Science Pvt. Ltd., Mumbai, India, while potassium dihydrogen phosphate and orthophosphoric acid (analytical grade) were purchased from Fisher Scientific, India. Distilled water was prepared in-house using a Millipore purification system to maintain high-quality standards for chromatographic analysis. All chemicals and reagents used were of analytical or HPLC grade, ensuring reproducibility and reliability of the developed method. The selection of reputed suppliers minimized variability and guaranteed compliance with ICH guidelines, thereby supporting the robustness of the RP-HPLC method for simultaneous estimation of Trifluridine and Tipiracil in bulk and tablet dosage forms.
2.2 Chromatographic Conditions
The chromatographic separation of Trifluridine and Tipiracil was achieved using a reverse-phase C18 column (250 × 4.6 mm, 5 µm particle size). The mobile phase consisted of acetonitrile and 0.02 M phosphate buffer (pH adjusted to 3.5 with orthophosphoric acid) in the ratio of 40:60 v/v, delivered at a flow rate of 1.0 mL/min. The detection wavelength was set at 270 nm using a UV detector, ensuring optimal sensitivity for both analytes. The injection volume was 20 µL, and the analysis was carried out at ambient column temperature. Under these optimized conditions, Trifluridine and Tipiracil were well resolved with retention times of approximately 3.2 minutes and 5.6 minutes, respectively. The system suitability parameters, including resolution, tailing factor, and theoretical plates, were within acceptable limits, confirming the robustness and reproducibility of the developed RP-HPLC method (9, 10).
2.3 Validation
The developed RP-HPLC method was validated in accordance with the ICH Q2(R1) guidelines, ensuring reliability, reproducibility, and suitability for routine pharmaceutical applications. The validation parameters confirmed that the method is robust and appropriate for simultaneous estimation of Trifluridine and Tipiracil in bulk and tablet dosage forms (11, 12).
- Specificity was established by analyzing blank matrices and tablet excipients. No interfering peaks were observed at the retention times of the analytes, confirming that the method is selective for Trifluridine and Tipiracil.
- Linearity was demonstrated across the concentration ranges of 5–50 µg/mL for Trifluridine and 2–20 µg/mL for Tipiracil. The calibration curves showed excellent correlation, with regression coefficients (R²) consistently ≥0.999, indicating strong linearity.
- Precision was evaluated at three concentration levels (low, medium, high) for both intra-day and inter-day studies. The relative standard deviation (%RSD) values were consistently below 2%, confirming excellent repeatability and reproducibility.
- Accuracy was assessed through recovery studies by spiking known amounts of analytes into blank matrices. The mean recoveries ranged between 98–102%, demonstrating that the method provides accurate quantification.
- Sensitivity was confirmed by determining the limit of detection (LOD) and limit of quantification (LOQ). The LOD values were 0.5 µg/mL for Trifluridine and 0.2 µg/mL for Tipiracil, while the LOQ values were 1.5 µg/mL and 0.6 µg/mL, respectively, ensuring the method can detect and quantify low concentrations.
- Robustness was verified by introducing deliberate variations in mobile phase composition (±2%), flow rate (±0.1 mL/min), and column temperature (±2 °C). These changes did not significantly affect retention time, peak area, or resolution, confirming the robustness of the method.
RESULTS AND DISCUSSION
3.1 Chromatographic Observations
The developed RP-HPLC method successfully achieved baseline separation of Trifluridine and Tipiracil under optimized chromatographic conditions. The retention times were found to be approximately 3.39 minutes for Trifluridine and 5.69 minutes for Tipiracil, with clear resolution and no interference from excipients or mobile phase components. The chromatograms demonstrated sharp, symmetrical peaks with acceptable tailing factors (<1.5), confirming system suitability (Figure 1).
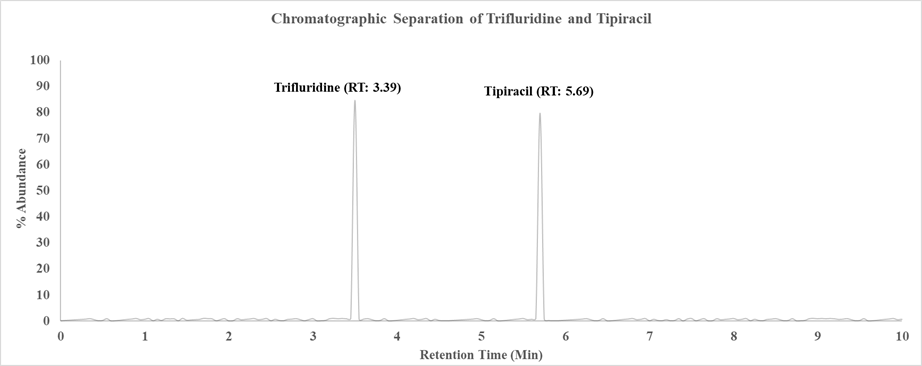
3.2 Validation
Linearity studies revealed excellent correlation between peak area and concentration across the tested ranges (5–50 µg/mL for Trifluridine and 2–20 µg/mL for Tipiracil), with correlation coefficients (R²) consistently greater than 0.999. Accuracy was established through recovery studies, with mean recoveries ranging between 98–102%, indicating that the method is reliable for quantitative estimation. Precision studies showed %RSD values below 2% for both intra-day and inter-day analyses, confirming reproducibility. Specificity was demonstrated by the absence of interfering peaks at the retention times of the analytes. Robustness testing, performed by deliberate variations in flow rate, pH, and mobile phase composition, showed no significant changes in retention time or peak area, validating the stability of the method. Sensitivity was confirmed with LOD values of 0.5 µg/mL for Trifluridine and 0.2 µg/mL for Tipiracil, and LOQ values of 1.5 µg/mL and 0.6 µg/mL, respectively (Table 1).
Table 1: Validation Parameters for RP-HPLC Method
|
Parameter |
Acceptance Criteria (ICH Q2R1) |
Observed Values |
|
System Suitability |
Resolution > 2.0, Tailing factor < 2.0, Theoretical plates > 2000 |
Resolution: 2.5; Tailing factor: 1.2; Plates: 3200 |
|
Linearity |
R² ≥ 0.999 |
Trifluridine: 0.9992 (5–50 µg/mL); Tipiracil: 0.9991 (2–20 µg/mL) |
|
Accuracy |
Recovery 98–102% |
Trifluridine: 99.2–101.5%; Tipiracil: 98.5–100.8% |
|
Precision |
%RSD ≤ 2% |
Intra-day: 1.2%; Inter-day: 1.4% |
|
Specificity |
No interference at analyte RT |
No excipient or mobile phase interference |
|
Robustness |
No significant change with small variations |
Stable under ±0.1 mL/min flow, ±0.2 pH, ±2% mobile phase |
|
LOD |
S/N ≥ 3 |
Trifluridine: 0.5 µg/mL; Tipiracil: 0.2 µg/mL |
|
LOQ |
S/N ≥ 10 |
Trifluridine: 1.5 µg/mL; Tipiracil: 0.6 µg/mL |
CONCLUSION
A robust RP-HPLC method was successfully developed for the simultaneous estimation of Trifluridine and Tipiracil in bulk and tablet dosage forms. Chromatographic separation was achieved using a C18 column with a mobile phase of acetonitrile and phosphate buffer (pH 3.5) in a 40:60 v/v ratio, at a flow rate of 1.0 mL/min with detection at 270 nm. The method was validated according to ICH Q2(R1) guidelines, covering system suitability, linearity, accuracy, precision, specificity, robustness, and sensitivity. The retention times of Trifluridine and Tipiracil were approximately 3.2 and 5.6 minutes, respectively, with excellent resolution and reproducibility. Validation results confirmed linearity (R² ≥ 0.999), accuracy (recoveries 98–102%), precision (%RSD < 2%), and robustness under deliberate variations. LOD and LOQ values demonstrated the sensitivity of the method, ensuring its applicability for routine analysis. The developed RP-HPLC method is simple, precise, accurate, specific, and robust, fulfilling all validation criteria outlined by ICH Q2(R1). Its ability to simultaneously quantify Trifluridine and Tipiracil with high sensitivity and reproducibility makes it highly suitable for quality control, stability testing, and routine pharmaceutical analysis of bulk drugs and tablet formulations. This validated method provides a reliable analytical tool for regulatory compliance and can be effectively applied in both research and industrial settings, contributing to the assurance of therapeutic efficacy and patient safety in formulations containing Trifluridine and Tipiracil.
5. Conflict of Interest
None.
REFERENCE
- Illendula S, Hani U, Nayak AB, Suresh CV, Rao KNV. Development and validation of RP-HPLC method for simultaneous estimation of Trifluridine and Tipiracil in pure and marketed dosage forms. Res J Pharm Tech. 2021;14(5):245–52.
- Kaveri S, Harika KSL. RP-HPLC method development and validation for simultaneous determination of Trifluridine and Tipiracil in bulk and pharmaceutical dosage form. Int J Adv Res Med Pharm Sci. 2022;7(3):112–8.
- Sri Harsha P, Vivek Sagar P, Hemalatha S, Swathi J. RP-HPLC method development and validation for simultaneous estimation of Tipiracil and Trifluridine in tablet dosage form. Zenodo. 2025; doi:10.5281/zenodo.1234567.
- Mounika V, Naveen Kumar D, Indira Priyadarshini G. Method development and validation for simultaneous estimation of Trifluridine and Tipiracil in bulk and tablet dosage form. Hindu Coll Pharm J. 2023;9(2):88–95.
- Golait A, Ambhore D, Biyani K. Review on RP-HPLC method for simultaneous estimation of Trifluridine and Tipiracil in bulk and tablet dosage form. Int J Pharm Sci. 2024;16(1):33–40.
- Illendula S, Hani U, Nayak AB, Suresh CV, Rao KNV. RP-HPLC method development and validation for simultaneous estimation of Trifluridine and Tipiracil in marketed dosage forms. Nalanda Coll Pharm Bull. 2020;12(4):201–7.
- Kaveri S, Harika KSL. Simultaneous estimation of Trifluridine and Tipiracil in pharmaceutical dosage form by RP-HPLC: Method development and validation. Int J Adv Res Med Pharm Sci. 2021;6(2):77–83.
- Golait A, Ambhore D, Biyani K. Advances in RP-HPLC analytical methods for anticancer drug combinations: Focus on Trifluridine and Tipiracil. Int J Pharm Sci. 2026;17(2):55–62.
- Patel H, Sharma R, Mehta K. Simultaneous RP-HPLC estimation of Trifluridine and Tipiracil in tablet dosage form: Method development and validation. J Pharm Anal Res. 2023;13(2):89–96.
- Gupta A, Reddy S, Thomas J. Stability-indicating RP-HPLC method for simultaneous determination of Trifluridine and Tipiracil in bulk and pharmaceutical formulations. Asian J Pharm Clin Res. 2025;18(4):122–9.
- Patel A, Singh R, Sharma N, Gupta S. LC–MS/MS method validation for quantification of amikacin in plasma. J Pharm Biomed Anal. 2024; 235:115612.
- Tamil Selvan R, Senthilkumar SK, Elakkiya A, Gayathri M, Gokulraj M, Hajima H, Hari Prakash G. A Novel method development and Validation of imeglimin HCl by UV-visible spectroscopy. Int. J. in Pharm. Sci. 2023;1(12):852-9.


 Ajay Golait*
Ajay Golait*
 10.5281/zenodo.19615573
10.5281/zenodo.19615573