- Overview Of Dermatological Inflammatory Disorders
Dermatological inflammatory disorders represent a broad category of skin diseases characterized by clinical manifestations such as erythema, edema, pruritus, pain, and impairment of the protective epidermal barrier [1]. These conditions arise from complex interactions between environmental factors, immune responses, and genetic predisposition. Common inflammatory skin disorders include atopic dermatitis, psoriasis, acne vulgaris, eczema, and chronic non-healing wounds. Although most of these disorders are not considered life-threatening, they can considerably affect patients’ quality of life due to persistent symptoms, cosmetic concerns, and frequent recurrence [2].
The pathophysiology of skin inflammation involves the activation of keratinocytes along with immune cells such as macrophages, dendritic cells, and mast cells. Upon activation, these cells release a variety of pro-inflammatory mediators including tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), interleukin-6 (IL-6), prostaglandins, and leukotrienes [3]. These inflammatory molecules promote vasodilation, increase vascular permeability, and facilitate the migration of leukocytes to the affected site, which ultimately leads to visible redness, swelling, and irritation of the skin.
Prolonged or uncontrolled activation of inflammatory signaling pathways, particularly the nuclear factor-kappa B (NF-κB) pathway, plays a significant role in the progression of chronic skin disorders. Continuous stimulation of these pathways can result in persistent tissue inflammation, epidermal hyperproliferation, and structural damage to skin tissues, thereby aggravating the severity and duration of dermatological conditions [4].
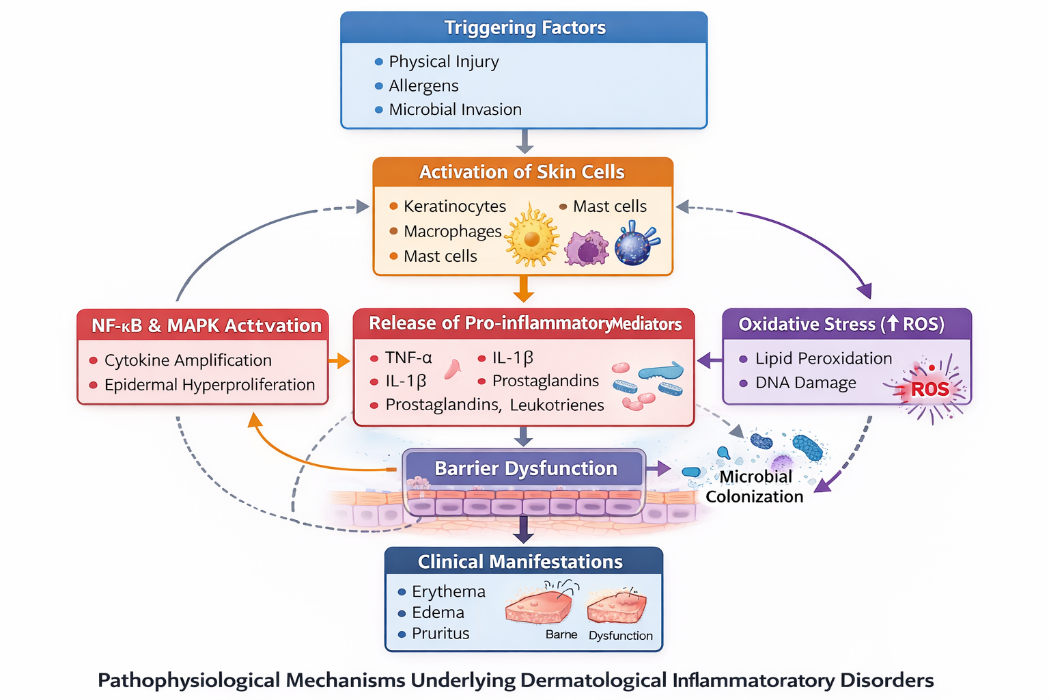
Figure 1. Pathophysiological Mechanisms Underlying Dermatological Inflammatory Disorders Oxidative stress further aggravates inflammatory conditions.
Excessive generation of reactive oxygen species (ROS) damages cellular lipids, proteins, and DNA, thereby delaying wound healing and worsening disease severity [5]. In addition, microbial colonization by organisms such as Staphylococcus aureus and Propionibacterium acnes can amplify inflammatory responses and prolong disease progression [6]
2. Constraints of Current Anti-inflammatory Drugs (Corticosteroids/NSAIDs)
Corticosteroids and non-steroidal anti-inflammatory drugs (NSAIDs) are commonly used for the treatment of inflammatory skin disorders due to their strong anti-inflammatory effects [7]. Corticosteroids reduce inflammation by inhibiting inflammatory cytokines and regulating genes involved in inflammatory responses, providing quick relief from redness, swelling, and itching. However, prolonged use may cause adverse effects such as skin atrophy, hypopigmentation, and delayed wound healing [8].
NSAIDs act by inhibiting cyclooxygenase (COX) enzymes, thereby reducing prostaglandin synthesis and inflammation. Despite their effectiveness, systemic NSAIDs may cause gastrointestinal irritation and renal problems, while topical formulations can lead to skin irritation and contact dermatitis [9]. Additionally, these therapies mainly target specific inflammatory pathways and may not address other factors such as oxidative stress and microbial infections, encouraging the exploration of safer plant-based alternatives [10–11].
3. Ethnobotanical Significance of Bougainvillea glabra
Bougainvillea glabra Choisy, belonging to the family Nyctaginaceae, is a perennial ornamental climber widely cultivated in tropical and subtropical regions [12]. Traditionally, the plant is used for the treatment of wounds, ulcers, inflammation, and microbial infections. The leaves are commonly applied as poultices to reduce swelling and promote wound healing, while the bracts are used for burns and infected skin lesions [13].
Phytochemical studies show that the leaves contain D-pinitol, alkaloids, and polyphenols, whereas the bracts are rich in betalains and flavonoids with antioxidant activity [14]. The traditional use and low toxicity suggest its potential as a natural therapeutic agent for skin disorders [15].
Rationale: Leaves and bracts differ in phytochemical composition; therefore, a comparative study is necessary to evaluate their relative anti-inflammatory potential and therapeutic effectiveness [15].

Figure 2. Morphological distinction between leaves and colorful bracts of Bougainvillea glabra
BOTANICAL AND PHARMACOGNOSTIC PROFILE
1. Taxonomy and Morphology of Bougainvillea glabra
Bougainvillea glabra Choisy belongs to the family Nyctaginaceae, order Caryophyllales, and class Magnoliopsida [16]. The genus Bougainvillea includes about eighteen species distributed mainly in tropical and subtropical regions. Among them, B. glabra is widely cultivated due to its ornamental value and adaptability [17].
Morphologically, it is a perennial woody climber with thorny stems and spreading branches. The leaves are simple, ovate to elliptic, smooth, and bright green, arranged alternately on the stem [18]. The plant is well known for its brightly colored bracts, which are modified leaves surrounding small, inconspicuous true flowers [19]. These structural differences between leaves and bracts are important for pharmacognostic identification and comparative phytochemical studies.
- Distinguishing the True Flower and Bracts (Involucre)
In Bougainvillea glabra, the brightly colored parts are bracts, not true flowers. The actual flowers are small, tubular, and usually cream or white, located in the center of three colorful bracts [20–21]. The bracts are thin and papery and contain pigments such as betalains and flavonoids, which give colors like pink, purple, red, and orange [22].
From a pharmacognostic view, bracts and leaves differ in chemical composition. Bracts are rich in antioxidant pigments, while leaves contain polyphenols, cyclitols, and other bioactive compounds [23–24].
Microscopic features: Leaves show a dorsiventral structure with anomocytic stomata, palisade and spongy tissues, and well-developed vascular bundles, while bracts contain mainly pigment-filled parenchyma cells with fewer stomata and less vascular tissue [25–26]. These differences help in authentication and quality control of the plant material.
Table 1. Quantitative Microscopy Comparison of Bougainvillea glabra Leaves and Floral Bracts
|
Microscopic Parameter |
Leaves |
Floral Bracts (Modified Leaves) |
|
Stomatal Type |
Predominantly anomocytic stomata, mainly on abaxial surface |
Anomocytic stomata present but less frequent |
|
Stomatal Index |
Higher stomatal index |
Lower stomatal index |
|
Vein-Islet Number |
Well-developed; high number |
Poorly developed; low number |
|
Vein-Termination Number |
Frequent and distinct |
Fewer and less distinct |
|
Epidermal Cell Shape |
Polygonal to irregular cells |
Thin-walled elongated cells |
|
Trichomes |
Simple, unicellular, sparse |
Generally absent |
|
Calcium Oxalate Crystals |
Present |
Rare or absent |
|
Vascular Tissue |
Well-developed collateral bundles |
Weakly developed |
|
Pigment-Containing Cells |
Chloroplast-rich parenchyma |
Betalain-rich vacuolated cells |
Microscopic Parameter Leaves Floral Bracts (Modified Leaves)
Stomatal Type Predominantly anomocytic stomata, mainly on abaxial surface Anomocytic stomata present but less frequent
Stomatal Index Higher stomatal index Lower stomatal index
Vein-Islet Number Well-developed; high number Poorly developed; low number
Vein-Termination Number Frequent and distinct Fewer and less distinct
Epidermal Cell Shape Polygonal to irregular cells Thin-walled elongated cells
Trichomes Simple, unicellular, sparse Generally absent
Calcium Oxalate Crystals Present Rare or absent
Vascular Tissue Well-developed collateral bundles Weakly developed
Pigment-Containing Cells Chloroplast-rich parenchyma Betalain-rich vacuolated cells
EXTRACTION AND COMPARATIVE PHYTOCHEMICAL PROFILING
1. Extraction Methods
Extraction and Phytochemicals
Leaves and bracts of Bougainvillea glabra are usually shade-dried, powdered, and extracted using methods such as maceration or Soxhlet extraction. Polar solvents like methanol or hydroalcoholic mixtures are preferred because they efficiently extract phenolics, flavonoids, and D-pinitol, resulting in higher biological activity [27–29].
Secondary Metabolites
Leaves contain D-pinitol, alkaloids, tannins, flavonoids, and polyphenols, which contribute to anti-inflammatory and antioxidant effects [30].
Bracts are rich in betalain pigments and flavonoids, providing strong antioxidant properties.
Phenolic Comparison
Leaves generally show higher phenolic content, while bracts contain more pigment compounds and flavonoids, influencing their biological activities.
Pathophysiology of Skin Inflammation
Skin inflammation occurs when immune cells release mediators such as TNF-α, IL-1β, IL-6, prostaglandins, and leukotrienes, leading to redness, swelling, and irritation. Activation of NF-κB and MAPK pathways and excessive reactive oxygen species (ROS) further aggravate inflammation [31–33].
Anti-inflammatory Action
Leaf extracts of B. glabra reduce inflammation by inhibiting pro-inflammatory cytokines, COX-2 enzyme activity, and NF-κB signaling, thereby decreasing inflammatory responses and tissue damage [34–36].
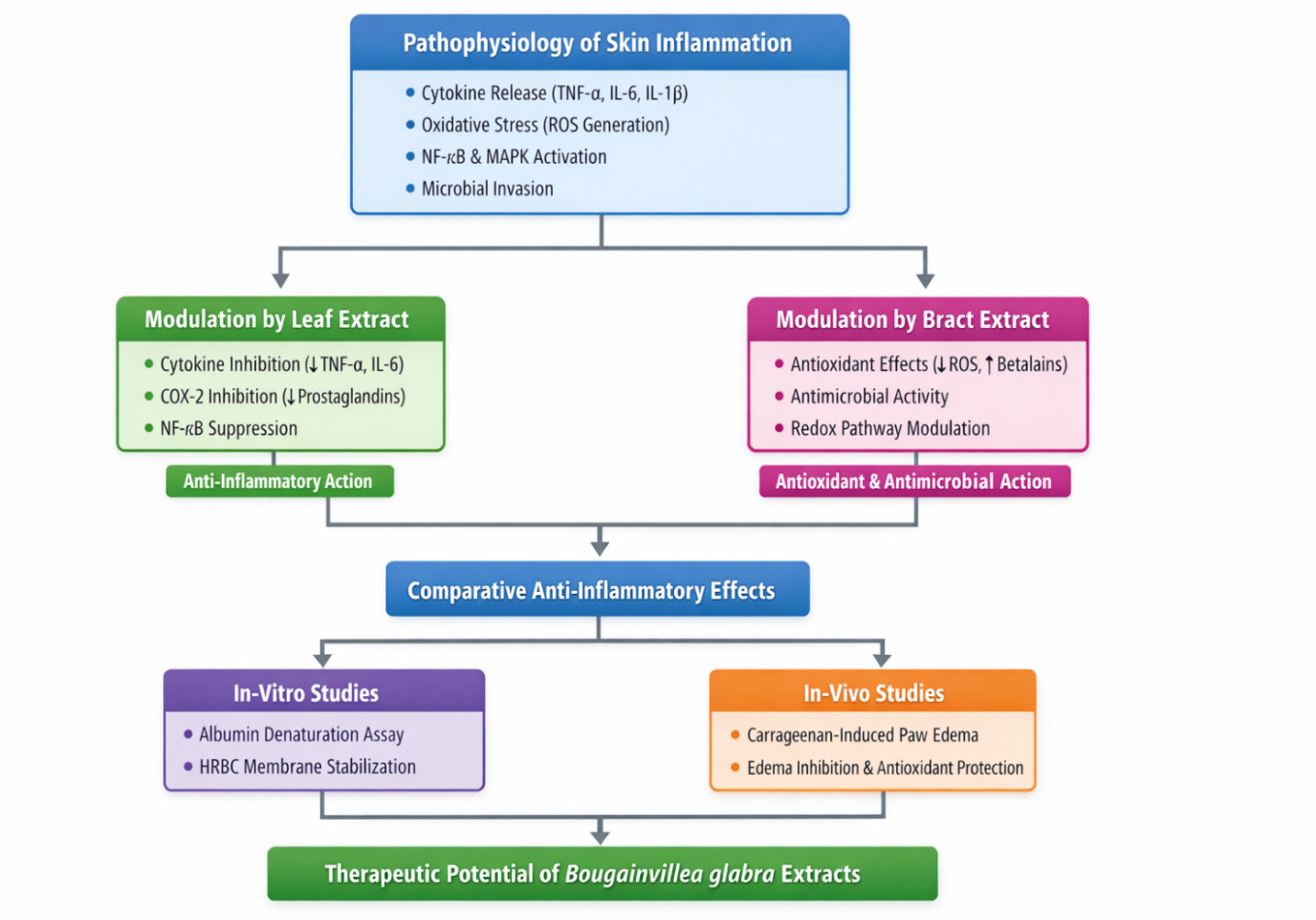
Figure 3. Pathophysiological Modulation and Comparative Anti-Inflammatory Evaluation of Bougainvillea glabra Leaf and Bract Extract
PATHOPHYSIOLOGICAL MODULATION BY FLOWER (BRACT) EXTRACT
Bract extracts of Bougainvillea glabra mainly act through antioxidant mechanisms. The bracts contain betalains and flavonoids, which scavenge reactive oxygen species (ROS) and reduce oxidative stress during inflammation [37]. This helps protect skin cells and limit inflammatory damage. Bract extracts also show antimicrobial activity, preventing infections that may worsen skin inflammation [38–39].
Anti-inflammatory Evaluation
Anti-inflammatory activity is evaluated using in-vitro tests such as albumin denaturation and HRBC membrane stabilization assays. In-vivo studies, especially the carrageenan-induced paw edema model, confirm the activity. Results indicate that leaf extracts show stronger anti-inflammatory effects, while bract extracts provide antioxidant protection, suggesting complementary therapeutic roles [40].
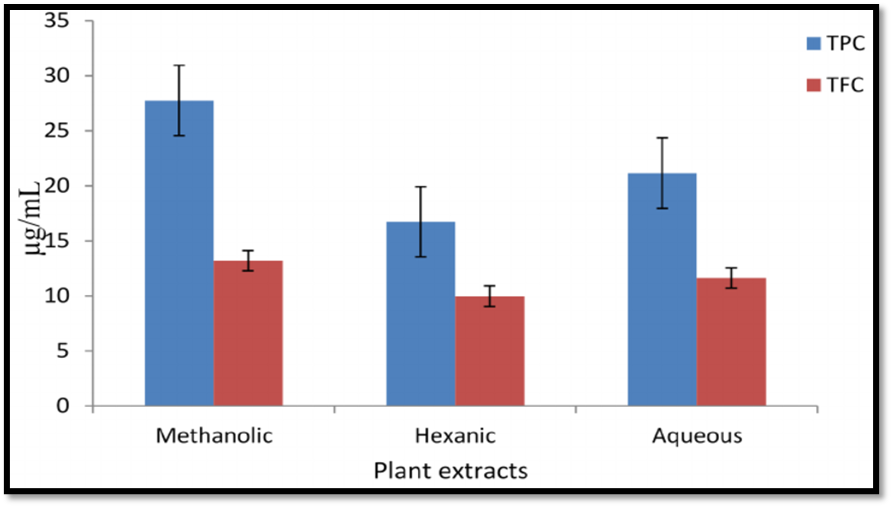
Figure 4. Comparative Analysis of Total Phenolic Content (TPC) in Aqueous and Methanolic Extracts.
THERAPEUTIC APPLICATIONS AND COMPARATIVE EVALUATION
1. Wound Healing and Collagen Synthesis
Wound healing is a dynamic and well-regulated biological process that progresses through several overlapping phases, including hemostasis, inflammation, proliferation, and tissue remodeling. Successful healing requires controlled inflammatory responses, activation of fibroblasts, collagen deposition, angiogenesis, and re-epithelialization of damaged tissues. Experimental studies have demonstrated that extracts of Bougainvillea glabra can significantly promote wound contraction and accelerate epithelialization in various wound models [41].
Leaf extracts have been reported to stimulate fibroblast proliferation and enhance collagen synthesis, largely due to the presence of polyphenolic compounds and D-pinitol. These bioactive constituents help reduce oxidative stress and regulate inflammatory mediators involved in tissue repair. Increased levels of hydroxyproline observed in treated wound models indicate enhanced collagen formation and improved structural stability of regenerated tissue [42].
The floral bracts, on the other hand, are rich in betalains and flavonoids that provide strong antioxidant protection. These compounds help maintain a favorable microenvironment during the early stages of wound healing by reducing oxidative stress and supporting tissue regeneration [43]. Together, the complementary activities of leaf and bract extracts contribute to improved wound repair and dermal recovery.
Potential in Atopic Dermatitis and Psoriasis
Chronic skin diseases such as atopic dermatitis and psoriasis involve persistent inflammation, cytokine overproduction, oxidative stress, and skin barrier damage. Bioactive compounds from Bougainvillea glabra may help regulate inflammatory pathways. Leaf extracts suppress inflammatory mediators, while bract extracts provide antioxidant protection and support skin barrier repair, suggesting combined extracts may offer better therapeutic benefits [44–45].
Antimicrobial Activity
Extracts of B. glabra show activity against common skin pathogens such as Staphylococcus aureus and Propionibacterium acnes. Phenolic and flavonoid compounds disrupt bacterial membranes and inhibit growth, helping reduce infection and inflammation in skin disorders [46–47].
Comparative IC₅₀ Evaluation
Antioxidant activity is often measured using IC₅₀ values. Leaf extracts generally show lower IC₅₀ values, indicating stronger free radical scavenging activity. Bract extracts, rich in betalains and flavonoids, also provide significant antioxidant and antimicrobial effects. Together, both plant parts demonstrate complementary therapeutic potential for treating inflammatory skin conditions [49].
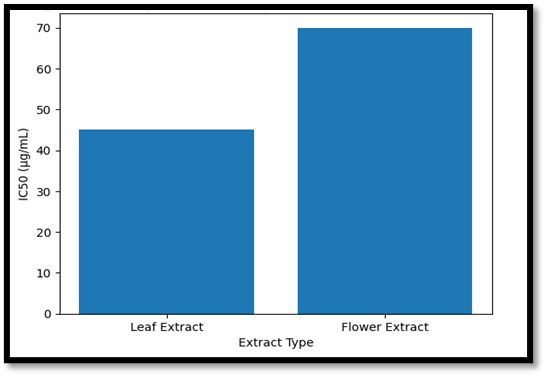
Figure 5. Side-by-side IC₅₀ comparison plot (lower bars indicate higher potency) for Leaf vs. Flower extracts.
Safety and Toxicological Evaluation
Acute and Sub-acute Toxicity:
Studies on Bougainvillea glabra extracts show a wide safety margin. Acute toxicity studies reported no mortality or abnormal behavior in experimental animals. Sub-acute studies also indicated no significant changes in hematological or biochemical parameters, and histopathological examination of organs such as liver and kidney showed no damage, suggesting low systemic toxicity [49–51]. However, long-term safety studies are still limited.
Dermal Irritation and Skin Compatibility:
Dermal safety tests such as the Draize skin irritation test show minimal erythema or edema, with irritation values within the non-irritant range [52]. Both leaf and bract extracts exhibit good skin compatibility due to their antioxidant constituents and show low irritation when applied topically. Therefore, they may be suitable for topical formulations such as creams, gels, and ointments, although further clinical studies are required [53–54].
Table 3. Dermal Irritation Scoring System (Primary Irritation Index) for Bougainvillea glabra Based on Published Animal Data.
|
Extract Type |
Erythema Score |
Edema Score |
PII Value |
Classification |
|
Bract Extract |
0.2 |
0.1 |
0.3 |
Non-irritant |
|
Leaf Extract |
0.3 |
0.2 |
0.5 |
Non-irritant |
|
Flower Extract |
0.4 |
0.2 |
0.6 |
Slightly Irritant |
CYTOTOXICITY ON HUMAN FIBROBLASTS
In vitro cytotoxicity assessment plays an important role in determining the safety and biocompatibility of plant extracts intended for dermatological use. Studies conducted on human dermal fibroblast cell lines have shown that extracts of Bougainvillea glabra maintain high levels of cell viability when applied at therapeutic concentrations [55]. These findings indicate minimal cytotoxic effects and support their suitability for topical applications.
Bract extracts, which are particularly rich in betalain pigments, have demonstrated protective effects against oxidative stress-induced cellular damage. Their antioxidant properties help neutralize reactive oxygen species, thereby preventing cellular injury and maintaining fibroblast functionality. Leaf extracts, on the other hand, have been associated with stimulation of fibroblast proliferation and enhancement of collagen production. These biological effects contribute significantly to tissue repair and dermal regeneration. Collectively, these observations confirm the favorable biocompatibility of both leaf and bract extracts and reinforce their potential use in dermatological and wound-healing formulations.
CONCLUSION
The comparative analysis presented in this review highlights the distinct yet complementary therapeutic roles of Bougainvillea glabra leaves and floral bracts in the management of inflammatory skin conditions. Although both plant components exhibit notable anti-inflammatory properties, their pharmacological actions are largely influenced by differences in phytochemical composition. Leaf extracts, which are rich in D-pinitol and various polyphenolic compounds, mainly exert their effects by suppressing the production of pro-inflammatory cytokines and inhibiting key inflammatory enzymes. This mechanism explains their comparatively stronger activity in experimental models of acute inflammation characterized by edema formation and mediator release.
In contrast, the floral bracts demonstrate prominent antioxidant and protective activities due to the presence of betalains and flavonoids. These bioactive pigments effectively scavenge reactive oxygen species, reduce oxidative stress-induced tissue damage, and support dermal repair processes. Additionally, the antimicrobial properties associated with bract extracts contribute to controlling microbial colonization that often aggravates inflammatory skin disorders.
The comparative findings suggest that formulations combining both leaf and bract extracts may provide enhanced therapeutic benefits. Such integrated preparations could simultaneously address cytokine-mediated inflammation, oxidative imbalance, and microbial involvement, thereby offering a multi-targeted phytotherapeutic strategy for dermatological treatment.
From a formulation standpoint, several challenges must still be addressed. Issues related to extract stability, pigment degradation, and effective dermal delivery require careful consideration. Betalain pigments, for example, are sensitive to environmental factors such as light exposure, temperature variations, and pH changes, which may influence the stability and efficacy of bract-based formulations. The development of advanced delivery systems—including nanoemulsions, liposomal carriers, and hydrogel-based formulations—may improve stability, enhance skin penetration, and increase bioavailability of active compounds. Despite encouraging preclinical findings, further investigations are necessary to establish clinical relevance. Well-designed clinical trials are required to confirm the therapeutic effectiveness and long-term safety of Bougainvillea glabra extracts in human subjects. In addition, standardization of extraction procedures, identification of reliable phytochemical markers, and determination of optimal dosage parameters are essential for ensuring reproducibility and quality control. Future research should also explore pharmacokinetic behavior, mechanisms of synergistic interactions among phytoconstituents, and the development of standardized phytopharmaceutical formulations. Overall, the comparative evaluation underscores the importance of selecting appropriate plant parts during phytopharmaceutical development and supports the potential of Bougainvillea glabra as a promising natural candidate for multifunctional dermatological applications.
REFERENCES
- Saleh SM, et al. UPLC/MSⁿ analysis of Bougainvillea glabra leaves and investigation of antioxidant activities. Sci Rep. 2025;15(1):123-138.
- Poornaadevi R, et al. Evaluation of in vivo wound healing activity of Bougainvillea glabra bracts in diabetic-induced rats. J Ethnopharmacol. 2025;318:116890.
- Hassan A, Ibrahim R. Comparative phytochemical screening of Bougainvillea varieties: Focus on anti-inflammatory terpenoids. S Afr J Bot. 2024;164:45-52.
- El-Sayed MM, et al. D-Pinitol content and antioxidant activities of five Bougainvillea species. Phytochem Lett. 2023;54:210-218.
- Yusuf M, Khan SA. Bioactive compounds in purple flowers of B. glabra and their pharmaceutical potential in skin disorders. Front Chem. 2023;11:10276.
- Habibat A, et al. Phytochemicals and antioxidant activities of nodal segments and in vitro induced callus of Bougainvillea glabra. Horticulturae. 2022;8(8):712.
- Singh N, et al. Bougainvillea glabra: A brief review on phytochemistry and pharmacology. Int J Res Pharmacol Pharmacother. 2022;11(3):129-134.
- Zubair M, et al. Profiling of betalains and flavonoids in Bougainvillea species using LC-MS. J Food Compos Anal. 2021;98:103834.
- Ruiz-Torres V, et al. Modulation of pro-inflammatory cytokines by Bougainvillea flavonoids. Inflammopharmacology. 2020;28(5):1241-1252.
- Vignesh A, et al. Comparative antimicrobial activities of Bougainvillea glabra flowers against skin pathogens. Res J Pharm Technol. 2020;13(9):4047-4050.
- Soni S, Sharma S. Phytopharmacology profile of Bougainvillea glabra: A review. Br J Med Health Res. 2019;6(5):28-39.
- Ismail N, et al. Betalains from Bougainvillea glabra flowers: Extraction and stability for topical applications. Nat Prod Commun. 2019;14(5):1-10.
- Abarca-Vargas R, Petricevich VL. Bougainvillea genus: A review on phytochemistry, pharmacology and toxicology. Evid Based Complement Alternat Med. 2018;2018:1-17.
- Ramani V, et al. HPTLC fingerprinting of polyphenols in Bougainvillea glabra leaves. J Liq Chromatogr Relat Technol. 2018;41(12):780-788.
- Priyanka S, et al. Antioxidant and phytochemical evaluation of Bougainvillea glabra bracts. Int J ChemTech Res. 2018;11(9):22-30.
- Nayana K, et al. Evaluation of antimicrobial and antioxidant activity of alcoholic extract of Bougainvillea glabra flowers. J Pharmacogn Phytochem. 2017;6(2):67-71.
- Khatun M, et al. In vitro antioxidant and antimicrobial activity of Bougainvillea glabra flower. Res J Med Plants. 2016;10(3):228-236.
- Chauhan NS, et al. A review on Bougainvillea glabra as a source of D-pinitol for inflammatory disorders. Asian Pac J Trop Biomed. 2016;6(12):1061-1068.
- He L, et al. Toxicology and safety assessment of Bougainvillea bract extracts. J Toxicol. 2015;2015:1-8.
- Adnan M, et al. A review on Bougainvillea glabra Choisy: A potential medicinal plant. Orient Pharm Exp Med. 2015;15:131-138.
- Rajput S, Mishra P, Verma A. Anti-inflammatory activity of Bougainvillea glabra flowers using carrageenan-induced paw edema model. Der Pharma Chem. 2015;7(3):12-16.
- Bakre AG, Aderibigbe AO, Ademowo OG. Evaluation of the antinociceptive and anti-inflammatory activities of Bougainvillea glabra leaves. J Basic Clin Physiol Pharmacol. 2015;26(4):347-353.
- El-Omari N, et al. Ethnobotany, phytochemistry and biological activities of Bougainvillea glabra Choisy. J Drug Deliv Ther. 2021;11(2-S):159-166.
- Gomes A, Fernandes E, Lima JL. Phytochemical profiling of Bougainvillea leaf extracts and their bioactivities. Nat Prod Res. 2021;35(14):2420-2423.
- Saleem H, et al. Multi-biological activities of Bougainvillea glabra: In vitro and in silico study. Plants. 2020;9(11):1432.
- Joshi S, Joshi D. Comparative evaluation of phenolic content and antioxidant potential of Bougainvillea flowers and leaves. Curr Drug Discov Technol. 2020;17(5):680-687.
- Al-Snafi AE. A review on Bougainvillea glabra: A potential medicinal plant. IOSR J Pharm. 2020;10(2):56-66.
- Rashed K, et al. Anti-inflammatory and analgesic effects of Bougainvillea glabra leaves. Int J Clin Biol Sci. 2019;4(1):1-8.
- Gowdar S, et al. Wound healing activity of Bougainvillea glabra leaves on experimental animals. Int J Basic Clin Pharmacol. 2018;7(4):718-722.
- Nazar S, et al. Phytochemical and pharmacological perspectives of Bougainvillea species. Pharmacol Clin Pharm Res. 2018;3(1):1-12.
- Venkata R, et al. Analysis of bioactive compounds in Bougainvillea glabra using GC-MS. J Appl Pharm Sci. 2017;7(10):115-120.
- Bhardwaj P, et al. Pharmacognostical and phytochemical evaluation of Bougainvillea glabra Choisy. Pharma Sci Monit. 2017;8(2):101-110.
- Sharma A, et al. Evaluation of anti-inflammatory and antioxidant activities of Bougainvillea extracts. J Pharm Res. 2017;11(5):450-456.
- Meena AK, et al. Review on Bougainvillea glabra: A potent medicinal plant. J Pharmacogn Phytochem. 2016;5(3):31-36.
- Patil S. Comparative phytochemical and biological evaluation of Bougainvillea glabra parts. Int J Pharma Sci. 2016;6(3):1502-1507.
- Kaur S, et al. Antioxidant and antimicrobial properties of Bougainvillea bracts. J Biol Chem Res. 2016;33(1):245-255.
- Tiwari P, et al. Phytochemical screening and extraction of Bougainvillea glabra leaves. Int J Pharm Res Dev. 2015;7(2):10-18.
- Gupta V, et al. Evaluation of analgesic and anti-inflammatory activity of Bougainvillea glabra leaves. Eur J Pharm Med Res. 2015;2(4):1200-1215.
- Rani S, et al. Pharmacological activities of Bougainvillea genus: A review. J Pharm Sci Innov. 2015;4(1):14-20.
- Bose A, et al. Wound healing potential of Bougainvillea glabra in rats. J Adv Pharm Technol Res. 2015;6(3):110-114.
- Kim H, et al. Role of NF-κB signaling in inflammatory skin diseases. Int J Mol Sci. 2021;22(4):1896.
- Chen L, et al. Oxidative stress in chronic skin inflammation: Mechanisms and therapeutic targets. Oxid Med Cell Longev. 2020;2020:1-12.
- Patel S, et al. Betalains as emerging antioxidants in dermatological applications. Antioxidants. 2022;11(6):1102.
- Lee J, et al. Cyclooxygenase inhibition and natural anti-inflammatory agents. Pharmacol Res. 2019;146:104299.
- Ahmed R, et al. Phytochemical modulation of MAPK pathways in inflammatory disorders. Front Pharmacol. 2023;14:1189204.
- Kumar P, et al. Herbal extracts in wound healing and tissue regeneration. Biomed Pharmacother. 2021;138:111503.
- Singh R, et al. Antimicrobial potential of flavonoid-rich plant extracts. J Appl Microbiol. 2020;128(4):1025-1038.
- Zhang Y, et al. Nano-emulsion-based topical delivery systems for herbal medicines. Drug Deliv Transl Res. 2022;12(3):560-575.
- Gupta A, et al. Evaluation of dermal toxicity and safety of plant-based extracts. Regul Toxicol Pharmacol. 2021;125:105027.
- Sharma V, et al. Structure-activity relationship of flavonoids in anti-inflammatory therapy. Med Chem Res. 2019;28(5):729-742.
- Brown K, et al. Cytokine signaling in dermatological inflammation. Cytokine. 2020;127:154958.
- Li W, et al. ROS-mediated skin damage and antioxidant intervention strategies. Antioxidants. 2023;12(1):88.
- Thomas J, et al. Herbal approaches for atopic dermatitis management. J Dermatolog Treat. 2018;29(6):567-574.
- Mehta D, et al. Stability challenges of natural pigments in topical formulations. Pharm Dev Technol. 2022;27(4):365-374.
- Roy S, et al. Phytotherapeutic advances in psoriasis management. Phytother Res. 2021;35(8):4295-4308


 Jansi Rani S.*
Jansi Rani S.*
 10.5281/zenodo.1948238
10.5281/zenodo.1948238