Myasthenia gravis (MG) is an autoimmune illness causing muscle weakness and exhaustion. The pathophysiology of MG is significantly influenced by autoreactive B cells that produce autoantibodies. The pathogenic autoantibodies in MG target elements of the post-synaptic muscle endplate at the neuromuscular junction, affecting neuromuscular transmission [17,19]. The disease is mainly caused by pathogenic antibodies directed against postsynaptic proteins, most commonly the acetylcholine receptor (AChR) and muscle-specific tyrosine kinase (MuSK), which lead to impaired neuromuscular transmission through receptor blockade, complement activation, and structural damage to the endplate [2,17,21]. According to epidemiological data, MG is becoming more common worldwide, which is indicative of better identification, advancements in diagnosis, and longer survival times [6].In addition to cytokine-mediated inflammation, autoreactive CD4⁺ T cells, B cells, plasmablasts, and plasma cells interact intricately in the immunopathogenesis of MG [9,10,14]. Dysfunction of regulatory T cells (Tregs), specifically decreased suppressive activity and altered FOXP3 expression, contributes to the breakdown of immunological tolerance [1,3-5]. Restoring Treg function can lessen the severity of the disease, according to experimental autoimmune myasthenia gravis (EAMG) models [5,15]. Autoreactive lymphocyte growth and ectopic germinal centre formation are facilitated by thymic abnormalities, such as thymic hyperplasia and thymoma-associated deficiencies in MHC class II and AIRE expression [4,18]. The percentage of seronegative disease cases has decreased due to advancements in diagnostic methods, particularly live and fixed cell-based assays (CBAs), which have greatly improved the detection of AChR, MuSK, a grin, and associated antibodies [7,13]. Susceptibility loci within HLA regions and immune regulatory genes have been identified through genetic investigations, including genome-wide association analyses, underscoring the heritable component of disease variability [28,31]. Conventional treatments, like immunosuppressants and corticosteroids, are still common but have side effects and a delayed onset [19,24]. B-cell depletion, complement inhibition, and BCMA-directed CAR-T cell therapy are examples of emerging molecular approaches that show promise clinical success and mark a paradigm change toward precision-based, cell- and gene-targeted therapies in MG [11,20].
PATHOGENESIS:
Myasthenia gravis (MG) is an autoimmune disorder dependent on T cells and mediated by antibodies, defined by complex relationships between CD4⁺ T lymphocytes and B cells, leading to the production of autoantibodies targeting neuromuscular junction (NMJ) proteins. Under normal circumstances, self-tolerance is maintained by peripheral tolerance mediated by regulatory T cells (Tregs), which inhibit excessive immune activation, and by central tolerance mechanisms in the thymus that eliminate autoreactive T cells [17]. Immune tolerance fails in MG due to Treg cell failure or reduced activity, permitting autoreactive CD4⁺ T cells to continue functioning and activate [17].
Although peripheral lymphoid organs may later continue to produce antibodies, the thymus plays an essential role in the pathophysiology of disease by acting as a location for autoreactive B-cell activation and germinal centre formation [17]. Coordinated interactions between important postsynaptic proteins that control AChR clustering and stability, such as muscle-specific kinase (MuSK), low-density lipoprotein receptor-related protein 4 (LRP4), agrin, and rapsyn, are essential for effective neuromuscular transmission [19].AChR clustering is disrupted, and the NMJ becomes structurally unstable as a result of autoantibodies that target these proteins interfering with their signalling pathways [19].IgG4 autoantibodies inhibit correct assembly of AChRs at the postsynaptic membrane in MuSK-associated MG by disrupting the MuSK–LRP4 connection without activating complement [19]. Similarly, agrin-mediated signalling is hampered by anti-LRP4 antibodies, which may also trigger complement pathways and cause additional harm.
The neuromuscular junction is a specialised synapse at which motor neurons convey messages to skeletal muscle fibres via the release of the neurotransmitter acetylcholine (ACh). Nicotinic acetylcholine receptors (AChRs) on the postsynaptic muscular membrane bind to ACh, which is released into the synaptic cleft when a nerve impulse reaches the presynaptic terminal. muscular contraction results from this contact, which depolarises the muscular membrane [17]. Pathogenic autoantibodies targeting postsynaptic proteins such as AChR and MuSK compromise the integrity of this mechanism in MG, leading to impaired neuromuscular transmission [2,17].
The development of autoantibodies against proteins involved with neuromuscular transmission is the defining feature of MG. A lower percentage of individuals have antibodies against MuSK or other NMJ proteins, including LRP4 and agrin, but 80–85% of patients have antibodies against Ach [2,29]. Through a number of methods, these antibodies reduce neuromuscular transmission. They first stop acetylcholine from attaching to its receptor, which stops signals from being transmitted. Second, the density of AChRs on the muscle membrane is decreased by antibody binding, which causes cross-linking and quicker internalisation and destruction of AChRs. Third, the postsynaptic membrane is harmed by complement activation brought on by the antibody [17,29].
An important part of the pathophysiology of MG is complementing activation. The membrane attack complex (MAC) is created when AChR antibody binding triggers the conventional complement cascade. The MAC lowers the quantity of functioning acetylcholine receptors and damages the postsynaptic membrane structurally [21].
ROLE OF T- CELLS:
By promoting the development of autoantibodies and controlling immunological responses, T cells are essential to the immunopathogenesis of myasthenia gravis (MG). MG is regarded as a T cell-dependent, B cell-mediated autoimmune illness in which B cells are stimulated to make pathogenic antibodies against neuromuscular junction proteins, including muscle-specific kinase (MuSK) and acetylcholine receptors (AChR), by autoreactive CD4⁺ T helper cells [9,17]. These interactions take place through costimulatory signalling and cytokine production, which promote B-cell activation, proliferation, and differentiation into autoantibody-secreting plasma cells [10,14].
The development of illness is affected by several T cell subsets. Patients with MG have higher levels of T helper 17 (Th17) cells, which generate pro-inflammatory cytokines like interleukin-17 (IL-17), which support the autoimmune response and cause inflammation [9]. Additionally, follicular helper T cells play a role by promoting the development of germinal centres and boosting the generation of antibodies by B cells in lymphoid organs and the thymus [9,14]. The autoimmune response in MG is maintained and amplified in part by these T cell subsets.
The inability of regulatory T cells (Tregs), which typically minimise autoreactive immune responses and preserve immunological tolerance, is a significant component of MG pathogenesis. Research has demonstrated that MG patients have decreased expression of regulatory molecules, including FOXP3 and compromised Treg suppressive action, which results in unchecked activation of autoreactive T and B cells [1,3,5]. The significance of T-cell–mediated immune modulation in MG is further highlighted by experimental autoimmune myasthenia gravis (EAMG) models, which show that restoring Treg activity can lessen the severity of the disease [5,15].
REGULATORY T- CELLS IN MYASTHENIA GRAVIS:
A subset of T lymphocytes, known as regulatory T cells (Tregs), is essential for maintaining immunological tolerance and preventing autoimmune reactions. The expression of CD4, CD25, and the transcription factor FOXP3, which is crucial for their growth and suppressive function, are typical characteristic of these cells [1,3]. Immune tolerance is compromised and autoimmunity against neuromuscular junction components develops in myasthenia gravis (MG) due to deficiencies in the quantity or function of regulatory T cells [1,14].
Treg Development and Features:
Regulatory T cells grow largely in the thymus and are known as natural Tregs, while additional inducible Tregs might emerge in peripheral organs in response to antigen stimulation. By avoiding excessive inflammatory reactions and inhibiting effector T cell activation, these cells preserve immunological homeostasis. Mutations or decreased expression of the transcription factor FOXP3, which is essential for Treg development and function, might result in compromised immune control and heightened vulnerability to autoimmune disorders [1,3]. Tregs reduce antigen-presenting cell activity, secrete anti-inflammatory cytokines, and engage in direct cell-to-cell contact, among other ways.
Mechanisms of Immune Suppression by Tregs:
Tregs use a variety of mechanisms to control immune responses. The release of immunosuppressive cytokines like interleukin-10 (IL-10) and transforming growth factor (TGF-β), which limit inflammatory T-cell responses and lower immunological activation, is one significant mechanism [9,14]. By producing inhibitory molecules like CTLA-4, which hinder the induction of antigen-presenting cells and lessen the stimulation of effector T cells, Tregs can also dampen immunological responses. Furthermore, by ingesting interleukin-2 (IL-2), Tregs can alter metabolic pathways and restrict the growth of autoreactive T cells [14]. Tregs are essential in avoiding autoimmune reactions through these combined pathways.
Treg Functional Defects in Myasthenia Gravis:
Numerous investigations have shown that Tregs have decreased suppressive function in MG patients, permitting autoreactive T and B cells to continue being active [3]. The functional ability of Tregs to suppress immunological responses is frequently compromised, even though the total number of circulating Tregs may not necessarily be considerably decreased [3,14]. Immune control may be compromised in MG patients due to decreased expression of FOXP3 and other regulatory molecules [1,3]. Impaired neuromuscular transmission can occur from autoreactive T cells stimulating B cells to generate antibodies against acetylcholine receptors and other neuromuscular junction proteins [9,17].
Interaction of Tregs with Other Immune Cells:
Tregs have intimate interactions with other immune system cells that are implicated in the pathophysiology of MG. They inhibit the generation of pathogenic antibodies by controlling the activity of B cells, follicular helper T cells and CD4⁺ helper T cells (9,14). There has been evidence of an imbalance between pro-inflammatory T-cell subsets, such Th17 cells and regulatory T cells, in MG patients. Pro-inflammatory cytokines are produced more frequently as a result of increased Th17 cell activity, which accelerates autoimmune reactions and the advancement of the disease (9). Thus, a key factor in the pathophysiology of MG is the disturbance of the equilibrium between regulatory and inflammatory immune cells.Studies on experimentally generated myasthenia gravis (EAMG) have shown that whereas Tregs from diseased animals exhibit decreased regulatory activity, Tregs from healthy persons may suppress autoimmune responses [5,15]. It has been demonstrated that adding functioning Tregs to experimental models lessens the severity of the disease, demonstrating the protective effect of these cells in regulating autoimmune reactions [5].
B CELLS AND AUTOANTIBODY SYNTHESIS IN MYASTHENIA GRAVIS:
Since myasthenia gravis is mostly caused by pathogenic autoantibodies that target neuromuscular junction (NMJ) components, B cells are essential to the disease's development (9,17). Autoreactive B cells in MG develop into plasmablasts and plasma cells that make antibodies against NMJ proteins after being stimulated by contact with CD4⁺ T helper cells (10,14). These autoantibodies contribute to the typical muscle weakness seen in MG patients by impairing neuromuscular transmission.
Activation of Autoreactive B Lymphocytes:
Antigen presentation and T-cell assistance in lymphoid organs and the thymus activate autoreactive B cells. The development of germinal centres, where B lymphocytes proliferate and undergo affinity maturation, is facilitated by follicular helper T cells [9,14]. After being stimulated, B cells undergo differentiation into plasmablasts and plasma cells that have the capacity to generate significant quantities of pathogenic autoantibodies [10].
Autoantibodies Directed Against Neuromuscular Junction Proteins:
While some MG patients develop antibodies towards muscle-specific kinase (MuSK), the protein LRP4, or agrin, most generate antibodies that target the acetylcholine receptor (AChR) [2,29]. By preventing receptor function, encouraging receptor degradation, and triggering complement-mediated destruction at the postsynaptic membrane, these autoantibodies impede neuromuscular transmission [17,21].
B-Cell Clone Persistence:
Research has shown that even after thymectomy, autoreactive B-cell clones that originated in the thymus may continue to circulate (4). In patients with MG, these long-lived plasma cells may contribute to incomplete remission or illness recurrence by continuing to manufacture autoantibodies [4,10]. This persistence emphasises how crucial it is to target B cells and plasma cells in contemporary treatment approaches [11].
CYTOKINES' FUNCTION IN MYASTHENIA GRAVIS:
Pro-Inflammatory Cytokines:
In MG, cytokines are crucial for controlling immunological responses. MG patients have been found to have higher amounts of pro-inflammatory cytokines, including interleukin-17, also known as (IL-17) and interleukin-21 (IL-21), which are linked to increased autoimmune activity [9]. In addition to mediating inflammation, cytokines control the survival and development of immune cells in MG. Patients with MG have been found to have elevated levels of interleukin-6 (IL-6), thereby encouraging autoimmune reactions and B-cell development into antibody-producing plasma cells [9,14].Interleukin-21 (IL-21), which is mostly generated by follicular helper T cells, is vital for the development of germinal centres and stimulates the production of high-affinity autoantibodies, which maintains the course of the disease[9,14].Another pro-inflammatory cytokine that is elevated in MG is tumor necrosis factor-alpha (TNF-α), which increases immune cell recruitment and damages neuromuscular junctions [9].
B-Cell Activation Mediated by Cytokines:
A proliferation-inducing ligand (APRIL) and B-cell activating factor (BAFF) are two examples of cytokines that support B-cell survival, proliferation, differentiation, and autoantibody synthesis, which advances the disease [9,11]. In addition, dysregulation of anti-inflammatory cytokines like IL-10 has been documented, indicating impaired regulatory mechanisms and ineffective inhibition of autoreactive immune responses[14].Maintaining chronic inflammation and illness persistence in MG is largely dependent on the imbalance between pro-inflammatory cytokines (IL-17, IL-6, TNF-α) and anti-inflammatory cytokines (IL-10, TGF-β) [9,14].Immune homeostasis is upset, and prolonged inflammation at the neuromuscular junction in MG is supported by an imbalance between pro-inflammatory and regulatory cytokines [9,14].
ABNORMALITIES OF THE THYMUS IN MYASTHENIA GRAVIS:
The thymus largely influences the development of MG, and many patients have thymic abnormalities, including thymoma or thymic hyperplasia [18,27]. The stimulation of autoreactive T and B lymphocytes involved in autoimmune reactions is facilitated by these anomalies.B-cell activation and the generation of autoantibodies towards neuromuscular junction proteins are facilitated by the presence of aberrant germinal centres in the thymus of MG patients [4,18].AIRE and the molecules of the major histocompatibility complex (MHC) are examples of immune regulatory genes whose aberrant expression may compromise central immunological tolerance and encourage the growth of autoreactive cells in the thymus [18,27]. Ectopic germinal centers, which resemble additional lymphoid organs, are frequently seen in the thymus of MG patients, allowing autoreactive B and T cells to be continuously activated [4,18]. Thymic epithelial cells that express the autoimmune regulator (AIRE) gene abnormally allow autoreactive T cells to avoid negative selection and enter the peripheral circulation[18,27]. Due to decreased expression of major histocompatibility complex (MHC) class II molecules, thymoma-associated MG has altered antigen presentation, which impairs central tolerance [18].Additionally, abnormal expression of neuromuscular junction proteins such AChR subunits, which may function as autoantigens and activate immune system responses, is seen in thymic epithelial cells in MG[27].The course of the disease is made worse by chronic inflammation in the thymus, which encourages cytokine release and maintains autoreactive lymphocyte activation[18,27]
GENETIC AND MOLECULAR FACTORS IN MYASTHENIA GRAVIS:
Genetic Susceptibility:
MG susceptibility is significantly influenced by genetic variables; multiple studies have found links between the disease and particular human leukocyte antigen (HLA) alleles [28,31]. These genetic differences may raise the likelihood of autoimmune reactions and affect immune control. Several susceptibility genes linked to MG have been found by genome-wide association studies (GWAS), especially in the HLA-DR and HLA-DQ regions, which affect immunological recognition and antigen presentation [28,31].Immune regulatory gene polymorphisms, such as those in CTLA-4 and PTPN22, have been associated with changes in T-cell activation and a greater chance of autoimmune reactions in MG patients [31].Gene expression patterns linked to immunological dysregulation in MG have also been linked to epigenetic changes including DNA methylation and histone acetylation [31].Different immune cell subpopulations with varied transcriptional profiles have been identified by single-cell RNA sequencing investigations, suggesting heterogeneity in disease causes and progression [12].In MG, changes in signaling pathways like JAK-STAT and NF-κB also contribute to autoantibody formation and persistent inflammation [31].
Molecular and Immune Pathways:
Immune-related genes implicated in T-cell activation, B-cell control, and inflammatory processes that influence MG pathogenesis have been found through molecular research [31]. Immune tolerance loss and increased autoantibody production can result from changes in these pathways.Latest single-cell RNA sequencing research has found illness-specific immune cell subsets linked to disease activity and demonstrated heterogeneity amongst immune cells in MG patients [12].
EMERGING CELL- AND GENE-BASED THERAPEUTIC STRATEGIES
B-Cell-Specific Therapies:
B-cell-targeted treatments aim to lower the pathogenic B-cell populations that cause Myasthenia gravis autoantibody production. To prevent antibody-mediated injury at the neuromuscular junction, these strategies mainly target mature B cells, memory B cells, and plasmablasts [11]. Long-lived plasma cells may endure; however, monoclonal antibodies such as anti-CD20 drugs reduce autoantibody levels by depleting circulating B cells. Targeting FcRn to increase IgG autoantibody breakdown and blocking survival factors like BAFF to impede B-cell maturation and function are two more tactics. Therapeutic monoclonal antibodies in myasthenia gravis reduce B-cells through mechanisms such as direct apoptosis, antibody-dependent cellular phagocytosis (ADCP), antibody-dependent cellular cytotoxicity (ADCC), and complement-dependent cytotoxicity (CDC), resulting in a reduction of pathogenic autoantibodies [11,21]. Therefore, focused treatment for MG can be achieved, and immunological dysregulation can be corrected by selectively targeting B cells. Treatments like anti-CD20 antibodies aim to lower pathogenic B-cell populations that produce autoantibodies, making targeting B cells a crucial therapeutic strategy [11].
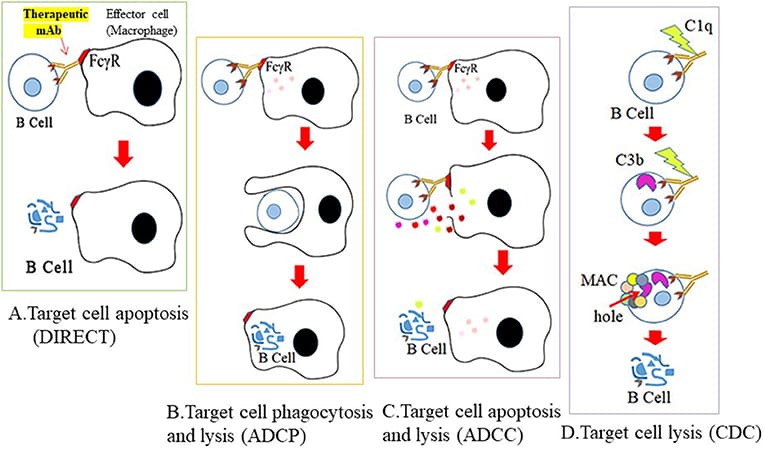
FIGURE-1| Mechanisms of therapeutic B cell destruction. There are four possible ways that therapeutic antibodies mediate their cytotoxic effects. Cross-linking of mAb to B cell surface antigen (A) inhibits and binds key receptors that mediate B cell survival (direct apoptosis), (B) stimulates engagement of effector cells through recognition of its Fc sequence and subsequent phagocytosis by effector cells (ADCP), (C) lysis by granzymes produced from effector cells (ADCC), or (D) activates the complement cascade and lysis by MAC formation on the cell surface and C3 deposits.
CAR-T Cell Treatment:
A comprehensive cell-based approach to addressing immunological dysregulation in myasthenia gravis is chimeric antigen receptor (CAR) T-cell therapy. By targeting B-cell maturation antigen (BCMA) expressed on antibody-producing plasma cells, modified T cells can selectively remove and reduce harmful autoantibodies [16,20,26]. Clinical research has shown that CAR-T therapy causes circulating B cells to be rapidly and persistently depleted, with lasting B-cell aplasia seen for several months following treatment [16]. Long-term regulation of humoral immunity is indicated by the slow recovery of B-cell populations over time, primarily as naïve B cells, whereas memory B cells and plasma cells continue to be repressed [16]. In addition, CAR-T treatment successfully reduces autoimmune responses by drastically lowering immunoglobulin levels and inflammatory activity. Its role in immune reprogramming is further supported by molecular investigations that show changes in the makeup of immune cells and a decrease in the expression of inflammatory genes after treatment [20].
All things considered; BCMA-directed CAR-T treatment presents a potentially revolutionary approach for refractory MG by selectively removing autoreactive plasma cells and restoring immunological balance [21].
CAR T-cell therapy targeting B-cell maturation antigen (BCMA) has shown promising results in patients with refractory MG by eliminating antibody-producing plasma cells [16,20,26].
Future Gene-Based Therapies:
By carefully altering immune pathways at the molecular level, future gene-based treatments aim to address immunological dysregulation in myasthenia gravis. These strategies focused on restoring immunological tolerance and controlling the genes that produce autoantibodies [22,24]. Using viral or non-viral vectors to deliver therapeutic genes that can improve regulatory processes or inhibit harmful immune responses is one method of gene therapy. In order to slow the progression of the disease, methods including gene editing (such as CRISPR-based approaches) are being investigated to specifically target autoreactive B cells or alter T-cell activity [24]. In addition, by altering the immune system to stop the production of autoantibodies, designed cell-based gene treatments, such as modified immune cells, may offer long-term control. Compared to traditional immunosuppressive medications, these cutting-edge medicines provide the possibility of long-term remission and more accurate, individualised treatment [22,24].
DISCUSSION
Myasthenia gravis (MG) is a complex autoimmune disease caused by abnormal interactions between T cells, B cells, and other immunological mediators. Pathogenic autoantibodies that target neuromuscular junction proteins are the main feature of the condition, which impairs synaptic transmission and weakens muscles. A growing body of research indicates that immunological tolerance deficiencies, namely those affecting regulatory T cells (Tregs) and their essential transcription factor FOXP3, are crucial in maintaining the autoimmune response.
Because B cells differentiate into plasma cells that continuously make autoantibodies against AChR, MuSK, and similar proteins, their contribution is equally important. For many individuals, incomplete remission can be explained by the persistence of long-lived plasma cells even after standard treatments like immunosuppression or thymectomy. Furthermore, by encouraging B-cell activation and persistent inflammation, pro-inflammatory cytokines like IL-17 and survival factors like BAFF exacerbate immunological dysregulation.
Targeted therapies have replaced widespread immunosuppression as the therapeutic focus due to recent developments in molecular immunology. Monoclonal antibodies and B-cell-depleting treatments have demonstrated therapeutic effects by lowering the number of autoreactive cells in circulation. By eradicating antibody-producing plasma cells and reprogramming immune responses, more sophisticated techniques like BCMA-directed CAR-T cell therapy provide a very targeted approach. In a similar vein, RNA-based CAR-T treatments offer a less dangerous option with temporary expression, reducing long-term side effects.
Another exciting area of research is gene-based treatments. Immune tolerance may be essentially restored by methods that modify autoreactive immune cells, restore FOXP3 expression, or alter cytokine signalling pathways. Additionally, advances like single-cell RNA sequencing have made it possible to identify immune cell subsets specific to a disease, opening the door for precision medicine techniques in MG.
CONCLUSION
Myasthenia gravis is a complex autoimmune disease that primarily affects T cells, B cells, and cytokine networks. It involves complicated immunological dysregulation. Although they offer symptomatic relief, conventional medicines are constrained by systemic adverse effects and delayed action. By directly treating the underlying immunological problems, emerging cell- and gene-based medicines provide a more focused and potentially curative approach. In order to achieve durable remission, strategies like B-cell depletion, CAR-T cell therapy that targets BCMA, and gene manipulation approaches that attempt to restore immunological tolerance show great promise. These methods aid in long-term immunological balance in addition to lowering pathogenic autoantibody levels.
REFERENCES
- Alahgholi-Hajibehzad M, Kasapoglu P, Jafari R, Rezaei N. The role of T regulatory cells in immunopathogenesis of myasthenia gravis: implications for therapeutics. Expert review of clinical immunology. 2015 Jul 3;11(7):859-70.
- Tsonis AI, Zisimopoulou P, Lazaridis K, Tzartos J, Matsigkou E, Zouvelou V, Mantegazza R, Antozzi C, Andreetta F, Evoli A, Deymeer F. MuSK autoantibodies in myasthenia gravis detected by cell-based assay—a multinational study. Journal of Neuroimmunology. 2015 Jul 15; 284:10-7.
- Thiruppathi M, Rowin J, Li Jiang Q, Sheng JR, Prabhakar BS, Meriggioli MN. Functional defect in regulatory T cells in myasthenia gravis. Annals of the New York Academy of Sciences. 2012 Dec;1274(1):68-76.
- Jiang R, Hoehn KB, Lee CS, Pham MC, Homer RJ, Detterbeck FC, Aban I, Jacobson L, Vincent A, Nowak RJ, Kaminski HJ. Thymus-derived B cell clones persist in the circulation after thymectomy in myasthenia gravis. Proceedings of the National Academy of Sciences. 2020 Dec 1;117(48):30649-60.
- Gertel-Lapter S, Mizrachi K, Berrih-Aknin S, Fuchs S, Souroujon MC. Impairment of regulatory T cells in myasthenia gravis: studies in an experimental model. Autoimmunity reviews. 2013 Jul 1;12(9):894-903.
- Bubuioc AM, Kudebayeva A, Turuspekova S, Lisnic V, Leone MA. The epidemiology of myasthenia gravis. Journal of medicine and life. 2021 Jan;14(1):7. Zhang M, Yue C, Fang J, Tang J, Chen C, Zhou C, Yang W, An X, Lin Q, Zhang J. Exploring the Role of B-Cell Biotherapy in Myasthenia Gravis from a Genetic Perspective: A Mendelian Randomisation Study. Molecular Neurobiology. 2026 Jan;63(1):115.
- Spagni G, Gastaldi M, Businaro P, Chemkhi Z, Carrozza C, Mascagna G, Falso S, Scaranzin S, Franciotta D, Evoli A, Damato V. Comparison of fixed and live cell-based assay for the detection of AChR and MuSK antibodies in myasthenia gravis. Neurology: Neuroimmunology & Neuroinflammation. 2022 Oct 21;10(1): e200038.
- Chahin N, Sahagian G, Feinberg MH, Stewart CA, Jewell CM, Kurtoglu M, Miljković MD, Vu T, Mozaffar T, Howard Jr JF. Twelve-month follow-up of patients with generalised myasthenia gravis receiving BCMA-directed mRNA cell therapy. MedRxiv. 2024 Jan 4:2024-01.
- Uzawa A, Kuwabara S, Suzuki S, Imai T, Murai H, Ozawa Y, Yasuda M, Nagane Y, Utsugisawa K. Roles of cytokines and T cells in the pathogenesis of myasthenia gravis. Clinical & Experimental Immunology. 2021 Mar;203(3):366-74.
- Stathopoulos P, Kumar A, Nowak RJ, O’Connor KC. Autoantibody-producing plasmablasts after B cell depletion identified in muscle-specific kinase myasthenia gravis. JCI insight. 2017 Sep 7;2(17):e94263.
- Huda R. New approaches to targeting B cells for myasthenia gravis therapy. Frontiers in immunology. 2020 Feb 21;11:240.
- Jin W, Yang Q, Peng Y, Yan C, Li Y, Luo Z, Xiao B, Xu L, Yang H. Single-cell RNA-Seq reveals transcriptional heterogeneity and immune subtypes associated with disease activity in human myasthenia gravis. Cell Discovery. 2021 Sep 14;7(1):85.
- Kim SH, Chung HY, Kim M, Kim SW, Shin HY. Development and Application of a Cell-Based Assay for Detecting Anti-Agrin Antibodies Associated With Myasthenia Gravis. Journal of Clinical Neurology (Seoul, Korea). 2025 Feb 24;21(2):105.
- Wu Y, Luo J, Garden OA. Immunoregulatory cells in myasthenia gravis. Frontiers in Neurology. 2020 Dec 15;11:593431.
- Souroujon MC, Aricha R, Feferman T, Mizrachi K, Reuveni D, Fuchs S. Regulatory T cell–based immunotherapies in experimental autoimmune myasthenia gravis. Annals of the New York Academy of Sciences. 2012 Dec;1274(1):120-6.
- Tian DS, Qin C, Dong MH, Heming M, Zhou LQ, Wang W, Cai SB, You YF, Shang K, Xiao J, Wang D. B cell lineage reconstitution underlies CAR-T cell therapeutic efficacy in patients with refractory myasthenia gravis. EMBO molecular medicine. 2024 Feb 26;16(4):966.
- Conti-Fine BM, Milani M, Kaminski HJ. Myasthenia gravis: past, present, and future. The Journal of clinical investigation. 2006 Nov 1;116(11):2843-54.
- Ströbel P, Chuang WY, Chuvpilo S, Zettl A, Katzenberger T, Kalbacher H, Rieckmann P, Nix W, Schalke B, Gold R, Müller‐Hermelink HK. Common cellular and diverse genetic basis of thymoma‐associated myasthenia gravis: role of MHC class II and AIRE genes and genetic polymorphisms. Annals of the New York Academy of Sciences. 2008 Jun;1132(1):143-56.
- Menon D, Barnett C, Bril V. Novel treatments in myasthenia gravis. Frontiers in neurology. 2020 Jun 30;11:538.
- Vu T, Durmus H, Rivner M, Shroff S, Ragole T, Myers B, Pasnoor M, Small G, Karam C, Vullaganti M, Peltier A. BCMA-directed mRNA CAR T cell therapy for myasthenia gravis: a randomized, double-blind, placebo-controlled phase 2b trial. Nature Medicine. 2026 Jan 9:1-1.
- Tuzun E, Scott BG, Goluszko E, Higgs S, Christadoss P. Genetic evidence for involvement of classical complement pathway in induction of experimental autoimmune myasthenia gravis. The Journal of Immunology. 2003 Oct;171(7):3847-54.
- Kaminski HJ, Sikorski P, Coronel SI, Kusner LL. Myasthenia gravis: the future is here. The Journal of Clinical Investigation. 2024 Jun 17;134(12).
- Damato V, Spagni G, Monte G, Woodhall M, Jacobson L, Falso S, Smith T, Iorio R, Waters P, Irani SR, Vincent A. Clinical value of cell-based assays in the characterisation of seronegative myasthenia gravis. Journal of Neurology, Neurosurgery & Psychiatry. 2022 Sep 1;93(9):995-1000.
- Iorio R. Myasthenia gravis: the changing treatment landscape in the era of molecular therapies. Nature Reviews Neurology. 2024 Feb;20(2):84-98.
- Cai Y, Han L, Zhu D, Peng J, Li J, Ding J, Luo J, Hong R, Wang K, Wan W, Xie C. A stable cell line expressing clustered AChR: a novel cell-based assay for Anti-AChR antibody detection in Myasthenia Gravis. Frontiers in Immunology. 2021 Jul 8;12:666046.
- Granit V, Benatar M, Kurtoglu M, Miljković MD, Chahin N, Sahagian G, Feinberg MH, Slansky A, Vu T, Jewell CM, Singer MS. Safety and clinical activity of autologous RNA chimeric antigen receptor T-cell therapy in myasthenia gravis (MG-001): a prospective, multicentre, open-label, non-randomised phase 1b/2a study. The Lancet Neurology. 2023 Jul 1;22(7):578-90.
- Yasumizu Y, Ohkura N, Murata H, Kinoshita M, Funaki S, Nojima S, Kido K, Kohara M, Motooka D, Okuzaki D, Suganami S. Myasthenia gravis-specific aberrant neuromuscular gene expression by medullary thymic epithelial cells in thymoma. Nature Communications. 2022 Jul 22;13(1):4230.
- Renton AE, Pliner HA, Provenzano C, Evoli A, Ricciardi R, Nalls MA, Marangi G, Abramzon Y, Arepalli S, Chong S, Hernandez DG. A genome-wide association study of myasthenia gravis. JAMA neurology. 2015 Apr;72(4):396-404.
- Vincent A, Waters P, Leite MI, Jacobson L, Koneczny I, Cossins J, Beeson D. Antibodies identified by cell‐based assays in myasthenia gravis and associated diseases. Annals of the New York Academy of Sciences. 2012 Dec;1274(1):92-8.
- Cruz PR, Huda S, López-Ruiz P, Vincent A. Use of cell-based assays in myasthenia gravis and other antibody-mediated diseases. Experimental neurology. 2015 Aug 1;270:66-71.
- Avidan N, Le Panse R, Berrih-Aknin S, Miller A. Genetic basis of myasthenia gravis–a comprehensive review. Journal of autoimmunity. 2014 Aug 1;52:146-53.


 Anagha K P*
Anagha K P*
 10.5281/zenodo.19630828
10.5281/zenodo.19630828