
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
Ranchi College of Pharmacy, Ranchi, Jharkhand 834003, India
The nervous system represents one of the most intricate and dynamic networks in living organisms, orchestrating a vast array of physiological processes, behavioral responses, and cognitive functions essential for survival and adaptation. This review delves into the multifaceted organization and operation of the nervous system, providing an in-depth examination of its structural framework, cellular composition, and underlying physiological mechanisms. Key components such as the central nervous system (CNS) and peripheral nervous system (PNS) are explored, with a particular focus on their roles in sensory perception, motor coordination, and homeostatic regulation. The interplay between neurons and glial cells, including astrocytes, oligodendrocytes, and microglia, is highlighted, emphasizing their collective contribution to neural signaling and maintenance. Advances in neuroscience have expanded our understanding of neural plasticity?the capacity of the nervous system to adapt to environmental changes and injury?and the molecular pathways governing synaptic transmission and regeneration. Genetic and environmental influences on neural development and function are also critically assessed, shedding light on their role in both normal physiology and pathophysiology. Furthermore, this review addresses a spectrum of nervous system disorders, from neurodegenerative diseases like Alzheimer?s and Parkinson?s to psychiatric conditions such as depression and schizophrenia, underscoring their complex etiologies and clinical manifestations. Emerging therapeutic strategies, including stem cell therapy, neuroprosthetics, and advanced neuroimaging techniques, are discussed in the context of their potential to revolutionize treatment paradigms and improve patient outcomes. This synthesis of current research and translational applications provides a comprehensive perspective on the nervous system's pivotal functions and its centrality to health and disease.
The nervous system represents a sophisticated biological network that fundamentally defines the operational capabilities of multicellular organisms. This intricate arrangement of specialized cells and tissues operates through precisely coordinated electrical and chemical signaling mechanisms to maintain homeostasis and facilitate organism-environment interactions. At its core, the nervous system comprises two main divisions: the central nervous system (CNS), encompassing the brain and spinal cord, and the peripheral nervous system (PNS), which includes cranial and spinal nerves extending throughout the body. The cellular foundation of neural function lies in neurons, highly specialized cells characterized by their distinctive morphology and electrochemical properties. These cells possess elaborate dendritic arbors for receiving inputs, a cell body containing essential molecular machinery, and axons capable of rapid signal propagation. Supporting these neurons are glial cells, including astrocytes, oligodendrocytes, and microglia in the CNS, and Schwann cells in the PNS, which maintain optimal conditions for neural function through various mechanisms such as myelination, metabolic support, and immune defence. Signal transmission within the nervous system occurs through both electrical and chemical mechanisms. Action pote ntials, generated through the coordinated activity of voltage-gated ion channels, propagate along axons as electrical signals. At synapses, these electrical signals trigger the release of neurotransmitters, which bind to specific receptors on target cells, thereby converting electrical signals into chemical messages that can either excite or inhibit subsequent neural activity. The functional organization of the nervous system exhibits remarkable hierarchical complexity. At the molecular level, neurotransmitter systems, including acetylcholine, dopamine, serotonin, and various neuropeptides, regulate specific aspects of neural function. These systems interact within neural circuits that process specific types of information, such as visual perception, motor control, or emotional responses. These circuits, in turn, are integrated into larger networks that coordinate complex behaviors and cognitive processes. The nervous system demonstrates remarkable plasticity, allowing for adaptation to environmental changes and learning from experience. This adaptability operates through multiple mechanisms, including synaptic modification, dendritic remodeling, and the integration of newly generated neurons in specific brain regions. Such plasticity underlies essential processes like memory formation, skill acquisition, and recovery from injury. Recent technological advances have revolutionized our understanding of nervous system function. Techniques such as optogenetics, which allows for precise control of specific neural populations using light, and advanced imaging methods that enable visualization of neural activity in real-time, have provided unprecedented insights into neural circuit operation. Additionally, the emergence of sophisticated computational models has enhanced our ability to understand how neural networks process information and generate behaviour. Dysfunction of the nervous system underlies numerous pathological conditions, ranging from neurodevelopmental disorders to neurodegenerative diseases. Understanding the fundamental principles of nervous system organization and function has crucial implications for developing therapeutic strategies for these conditions. Current research focuses on various approaches, including stem cell therapy, gene editing, and development of novel pharmacological agents targeting specific neural circuits or molecular pathways. This intricate biological system continues to be a frontier of scientific investigation, with new discoveries regularly emerging about its organization and function. As our understanding deepens, particularly through the application of emerging technologies and analytical approaches, we gain increasingly sophisticated insights into how the nervous system enables organisms to perceive, respond to, and interact with their environment while maintaining internal homeostasis and supporting complex cognitive processes [1, 2].
? ? ? ?
? ? ? ? ? ? 
? ? ? ?
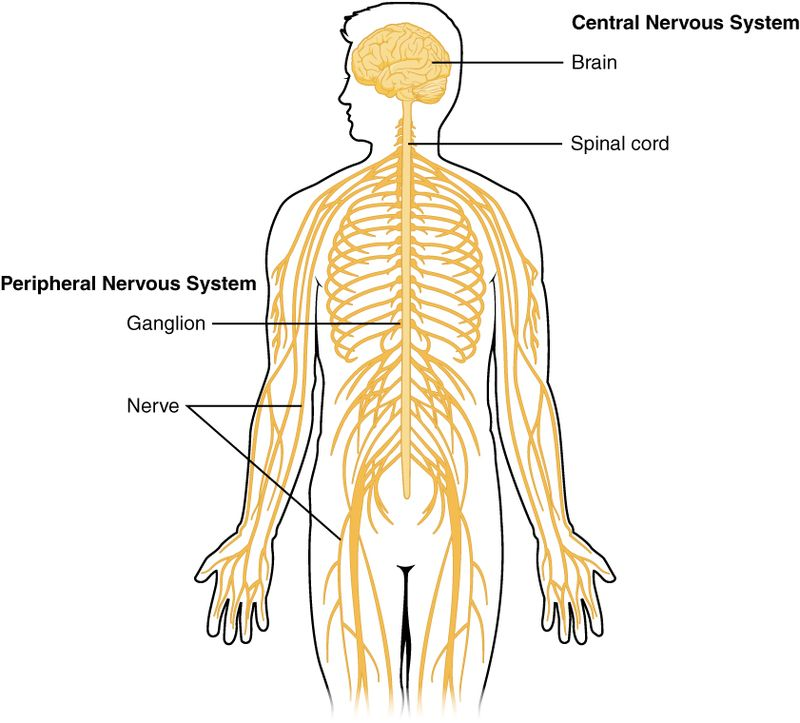
Fig. 1. Nervous System
The nervous system is divided into two major components: the central nervous system (CNS) and the peripheral nervous system (PNS). Each plays a distinct yet interconnected role in maintaining physiological and behavioral functions.
The central nervous system (CNS) represents the primary control center of the body, consisting primarily of the brain and spinal cord. This sophisticated network processes and integrates information from throughout the body, coordinating responses and maintaining homeostasis.
The CNS is encased within protective structures: the skull (cranium) houses the brain, while the vertebral column protects the spinal cord. Three layers of meninges ? the dura mater, arachnoid mater, and pia mater ? provide additional protection and support. Between the arachnoid and pia mater lies the subarachnoid space, filled with cerebrospinal fluid (CSF), which acts as a shock absorber and helps maintain optimal chemical conditions for neural function.
The brain, weighing approximately 1.4 kilograms in adults, consists of several major regions:
The cerebrum, the largest portion, is divided into two hemispheres connected by the corpus callosum. Its outer layer, the cerebral cortex, contains six distinct layers of neurons and is organized into four main lobes:
The diencephalon, located beneath the cerebrum, contains the thalamus (sensory relay station) and hypothalamus (homeostatic control center). The brainstem, comprising the midbrain, pons, and medulla oblongata, controls vital functions like breathing, heart rate, and blood pressure.
The cerebellum, positioned posterior to the brainstem, coordinates movement, balance, and motor learning. Its highly folded surface contains more neurons than the rest of the brain combined, despite occupying only 10% of total brain volume.
The spinal cord, a crucial component of the central nervous system, extends caudally from the medulla oblongata to the lumbar region. Functioning as a vital conduit, it houses neural pathways that facilitate communication between the brain and the peripheral nervous system. Upon cross-sectional examination, the spinal cord exhibits a distinct structural organization. Gray matter, characterized by the presence of neuronal cell bodies, occupies the central region, often described as a butterfly-shaped configuration. Surrounding the gray matter lies the white matter, composed of densely packed myelinated axons. These axons are meticulously organized into ascending and descending tracts, responsible for transmitting sensory and motor information, respectively. Furthermore, a central canal traverses the longitudinal axis of the spinal cord, containing cerebrospinal fluid (CSF) which provides vital support and protection to the delicate neural tissues.
The CNS comprises two main cell types:
The CNS operates through various integrated systems:
This comprehensive blood supply network is vital for the CNS to meet its high metabolic demands and sustain its critical functions, including sensory perception, motor control, and cognitive processes. The blood-brain barrier strictly regulates substance exchange between blood and neural tissue, maintaining a stable environment for optimal neural function.
The complex organization and function of the CNS enable sophisticated information processing and behavioral control, making it essential for survival and higher cognitive functions. Ongoing research continues to reveal new aspects of CNS function and potential therapeutic approaches for neurological disorders.
This intricate system's proper functioning is crucial for maintaining health and well-being, while its dysfunction can lead to various neurological and psychiatric conditions. Understanding its structure and function remains a central focus of neuroscience research and medical practice [3-7].
The Peripheral Nervous System (PNS) functions as a complex network of neural tissues that establishes vital connections between the central nervous system and various body structures. This sophisticated system comprises nerves, ganglia, and specialized receptors that extend throughout the organism, facilitating bidirectional information flow between central processing centers and peripheral targets.
Structurally, the PNS consists of two major functional divisions: the somatic nervous system and the autonomic nervous system. The somatic component primarily governs voluntary muscular activities and processes sensory information from the external environment. It encompasses both afferent pathways, which transmit sensory data from peripheral receptors to the CNS, and efferent pathways, which relay motor commands from the CNS to skeletal muscles. These neural circuits enable precise control of voluntary movements and facilitate rapid responses to environmental stimuli. The autonomic nervous system, conversely, regulates involuntary physiological processes and maintains homeostatic balance. This system further subdivides into sympathetic and parasympathetic branches, which generally exert opposing effects on target organs. The sympathetic division, often associated with "fight-or-flight" responses, utilizes norepinephrine as its primary neurotransmitter and increases physiological arousal. It accelerates heart rate, dilates bronchioles, inhibits digestive processes, and mobilizes energy reserves. The parasympathetic division, mediated primarily by acetylcholine, promotes "rest-and-digest" functions, reducing heart rate, facilitating digestion, and conserving energy.
At the cellular level, PNS neurons exhibit distinct structural and functional characteristics. Their cell bodies cluster in ganglia located outside the CNS, while their axons, often bundled into nerves, extend considerable distances to reach target tissues. These neurons utilize various neurotransmitters and neuromodulators to communicate with effector organs and other neural elements. Schwann cells provide myelin insulation for PNS axons, enhancing signal conduction velocity and maintaining axonal integrity. The PNS also contains specialized sensory receptors that detect specific environmental stimuli. These include mechanoreceptors for touch and pressure, thermoreceptors for temperature, nociceptors for pain, and proprioceptors for body position. These receptors transduce physical or chemical stimuli into electrical signals, which travel along sensory neurons to the CNS for processing. PNS function relies heavily on precise molecular mechanisms governing axonal transport, neurotransmitter synthesis and release, and receptor-mediated signaling. These processes ensure efficient communication between the CNS and peripheral tissues, enabling rapid and appropriate responses to both internal and external changes. Additionally, the PNS demonstrates remarkable regenerative capacity following injury, unlike the CNS, due to the supportive environment provided by Schwann cells and the expression of growth-promoting factors.
Understanding PNS organization and function proves crucial for medical practice, particularly in diagnosing and treating peripheral neuropathies, autonomic disorders, and neuromuscular conditions. Recent research continues to reveal new aspects of PNS function, including its role in neuroimmune interactions and its potential for therapeutic intervention in various pathological conditions [8-10].
Table 1. Overview of the Structural Organization of the Nervous System
|
Component |
Subcomponent |
Description |
|
Central Nervous System (CNS) |
Anatomical Organization |
Comprises the brain and spinal cord, protected by the skull, vertebral column, meninges, and cerebrospinal fluid (CSF). |
| ? |
Brain Structure and Components |
Includes the cerebrum (divided into lobes: frontal, parietal, temporal, occipital), diencephalon (thalamus, hypothalamus), brainstem (midbrain, pons, medulla), and cerebellum. |
| ? |
Spinal Cord Organization |
Features a butterfly-shaped gray matter core surrounded by white matter, organized into ascending (sensory) and descending (motor) tracts. |
| ? |
Neural Tissue Components |
Composed of neurons (signal transmitters) and glial cells (support and maintenance), including astrocytes, oligodendrocytes, microglia, and ependymal cells. |
| ? |
Functional Systems |
Sensory, motor, integrative, and communication systems for perception, voluntary movement, and autonomic regulation. |
| ? |
Blood Supply and Metabolism |
Supported by the internal carotid arteries, vertebral arteries, and Circle of Willis; protected by the blood-brain barrier. |
| ? |
Protection Mechanisms |
Includes physical protection (skull, vertebrae, meninges, CSF) and chemical barriers (blood-brain barrier, immune response). |
| ? |
Development and Plasticity |
CNS exhibits neuroplasticity for learning, adaptation, and recovery from injuries. |
| ? |
Communication Systems |
Utilizes electrical action potentials, neurotransmitters, and gap junctions for rapid and precise signaling. |
|
Peripheral Nervous System (PNS) |
Somatic Nervous System |
Controls voluntary muscular activities; includes afferent (sensory) and efferent (motor) pathways for communication with skeletal muscles. |
| ? |
Autonomic Nervous System |
Regulates involuntary functions; divided into sympathetic ("fight-or-flight") and parasympathetic ("rest-and-digest") divisions. |
| ? |
Neuronal Organization |
Neurons in ganglia outside the CNS; Schwann cells provide myelin for enhanced signal conduction. |
| ? |
Sensory Receptors |
Specialized receptors (e.g., mechanoreceptors, thermoreceptors, nociceptors) transduce environmental stimuli into electrical signals for CNS processing. |
| ? |
Regenerative Capacity |
Exhibits high regenerative ability due to Schwann cells and growth-promoting factors, unlike the CNS. |
? ? ? ?
? ? ? ? ? ? 
? ? ? ?
Fig. 2. Human Brain
? ? ? ?
? ? ? ? ? ? 
? ? ? ?
Fig. 3. Human Spinal Cord
The nervous system consists of two primary cell types: neurons and glial cells.
Neurons represent the fundamental cellular units of the nervous system, orchestrating the transmission of information through both electrical and chemical signaling mechanisms. These specialized cells exhibit distinct morphological and functional characteristics that enable complex neural processing.
The neuronal architecture comprises three primary components. The cell body, or soma, houses the nucleus and essential organelles responsible for protein synthesis and cellular maintenance. Dendrites extend from the soma like branching trees, creating elaborate networks that receive incoming signals from neighboring neurons. The axon, a specialized projection, conducts electrical impulses away from the soma toward target cells, often extending considerable distances through neural tissue. Each neuron possesses unique structural modifications that optimize its function. The axon terminal contains synaptic vesicles filled with neurotransmitters, while many axons feature myelin sheaths that enhance signal conduction velocity. Dendritic spines increase the surface area available for synaptic connections, facilitating robust neural networks.
Neurons can be categorized into three main functional classes based on their roles in neural circuits: Sensory neurons, also known as afferent neurons, specialize in detecting environmental stimuli and converting them into electrical signals. These neurons feature specialized receptor endings that respond to specific stimuli such as pressure, temperature, or chemical changes. They transmit this information from the periphery to the central nervous system for processing.
Motor neurons, or efferent neurons, conduct commands from the central nervous system to effector organs, primarily muscles and glands. These neurons typically have long axons and forms specialized junctions called motor end plates with muscle fibers, enabling precise control of movement and secretion.
Interneurons serve as intermediate processors within neural circuits, forming complex networks that integrate information between sensory and motor pathways. These neurons are typically multipolar, with extensive dendritic arbors that enable them to receive input from multiple sources and modify neural signalling patterns.
Neurons communicate through both electrical and chemical mechanisms. Action potentials, rapid changes in membrane potential, propagate along axons through the coordinated action of voltage-gated ion channels. At synapses, electrical signals trigger the release of neurotransmitters, which bind to specific receptors on target cells to continue signal propagation. Synaptic transmission involves complex molecular machinery that ensures precise signal transfer. Calcium-dependent exocytosis releases neurotransmitters into the synaptic cleft, while postsynaptic receptors and ion channels mediate the target cell's response. This process is highly regulated and can be modified through various molecular mechanisms, providing the basis for synaptic plasticity and learning.
The intricate organization and specialization of neurons enable the nervous system to process information with remarkable speed and precision. Understanding neuronal structure and function continues to reveal new insights into neural computation and the basis of behavior, while also providing crucial knowledge for treating neurological disorders [11-15].
Glial cells, also known as neuroglia, constitute a diverse population of non-neuronal cells that play crucial roles in maintaining optimal nervous system function. These cells, which outnumber neurons by approximately 10:1, perform essential supportive and regulatory functions throughout the central and peripheral nervous systems. Astrocytes represent the most abundant glial cell type in the central nervous system. These star-shaped cells extend numerous processes that contact both blood vessels and neurons, forming the foundation of the blood-brain barrier through their end-feet processes. Astrocytes regulate extracellular ion concentrations, particularly potassium, and maintain neurotransmitter homeostasis by removing excess neurotransmitters from synaptic clefts. They also provide metabolic support to neurons by supplying glucose and lactate, and participate in synaptic function through the release of gliotransmitters.
Oligodendrocytes serve as the primary myelinating cells within the central nervous system. These specialized cells extend multiple processes that wrap around axons, forming concentric layers of myelin membrane. Each oligodendrocyte can myelinate multiple axon segments simultaneously, with some capable of maintaining up to 40 separate myelin segments. This myelin sheath increases axonal conduction velocity through saltatory conduction and provides metabolic support to the axon. In the peripheral nervous system, Schwann cells perform the myelination function. Unlike oligodendrocytes, each Schwann cell myelinates only one axonal segment. These cells also play crucial roles in axon regeneration following injury by forming Bands of B?ngner, which guide regenerating axons to their targets. Microglia function as the resident immune cells of the central nervous system. These highly mobile cells continuously survey their environment for signs of injury or infection. Upon activation, microglia undergo morphological changes, becoming more amoeboid, and initiate inflammatory responses through cytokine release. They also participate in synaptic pruning during development and in response to injury, helping maintain neural circuit integrity.
Ependymal cells form a simple columnar epithelium lining the ventricular system of the brain and the central canal of the spinal cord. These ciliated cells contribute to cerebrospinal fluid production and circulation through their coordinated ciliary beating. They also form a selective barrier between the cerebrospinal fluid and brain parenchyma, regulating molecular exchange. Recent research has revealed increasingly complex roles for glial cells in nervous system function. These cells participate in synaptic plasticity, neural development, and disease processes. Astrocytes demonstrate calcium signaling capabilities and can modulate synaptic transmission. Oligodendrocytes exhibit remarkable plasticity in their myelinating capacity, responding to neural activity patterns. Microglia contribute to learning and memory processes through their synaptic pruning activities. Dysfunction of glial cells contributes to various neurological disorders. Astrocytic abnormalities appear in epilepsy and neurodegenerative diseases. Oligodendrocyte dysfunction characterizes demyelinating disorders like multiple sclerosis. Microglial activation patterns influence neuroinflammatory conditions and neurodegenerative disease progression. Understanding glial cell biology continues to reveal new therapeutic targets for neurological disorders, highlighting their essential role in nervous system health and disease [16-20].
Table 2. Cellular Components of the Nervous System
|
Cell Type |
Description |
Key Functions |
Structural Features |
Role in Disease |
|
Neurons |
Fundamental units of the nervous system responsible for information transmission. |
Signal transduction (electrical and chemical), processing information, and generating responses. |
Soma (cell body), dendrites, axon, axon terminal, myelin sheath. |
Neurodegenerative diseases (Alzheimer's, Parkinson's), epilepsy. |
|
Glial Cells |
Supportive cells that maintain optimal nervous system function. |
Support, protection, and modulation of neuronal activity. |
Diverse morphologies, including astrocytes, oligodendrocytes, microglia, and ependymal cells. |
Demyelinating diseases (multiple sclerosis), neuroinflammatory diseases. |
|
Astrocytes |
Most abundant glial cells. |
Blood-brain barrier formation, ion homeostasis, neurotransmitter uptake, metabolic support. |
Star-shaped with numerous processes. |
Epilepsy, neurodegenerative diseases. |
|
Oligodendrocytes |
Myelinating cells in the CNS. |
Axonal myelination, increasing conduction velocity. |
Multiple processes that wrap around axons. |
Demyelinating diseases. |
|
Microglia |
Immune cells of the CNS. |
Immune surveillance, phagocytosis, synaptic pruning. |
Highly mobile and phagocytic. |
Neuroinflammatory diseases, neurodegenerative diseases. |
|
Ependymal Cells |
Line the ventricles of the brain and central canal of the spinal cord. |
Cerebrospinal fluid production and circulation. |
Ciliated epithelial cells. |
Hydrocephalus. |
? ? ? ?
? ? ? ? ? ? 
? ? ? ?
Fig. 4. Neuron
? ? ? ?
? ? ? ? ? ? 
? ? ? ?
Fig. 5. Glial cell
Neurons employ sophisticated mechanisms of electrical and chemical signaling to transmit information throughout the nervous system. This communication forms the foundation of all neural processes, from basic reflexes to complex cognitive functions.
The action potential represents the fundamental unit of electrical signaling in neurons. This process initiates when the membrane potential reaches threshold through the sequential activation of voltage-gated ion channels. Initially, voltage-gated sodium channels open, allowing Na+ ions to flow into the cell, causing rapid membrane depolarization. This is followed by the delayed opening of voltage-gated potassium channels, leading to K+ efflux and membrane repolarization. The process concludes with a brief hyperpolarization period before returning to resting potential. Action potential propagation occurs through local circuit currents and saltatory conduction in myelinated axons. Myelin sheaths, produced by oligodendrocytes in the CNS, create high-resistance segments interrupted by nodes of Ranvier, where ion channels concentrate. This arrangement significantly increases conduction velocity while conserving metabolic energy.
Synaptic transmission is a fundamental process for neuronal communication. It commences when an action potential, a rapid electrical signal, arrives at the presynaptic terminal. This depolarization event triggers the opening of voltage-gated calcium channels, allowing calcium ions to influx into the presynaptic terminal. The elevated intracellular calcium concentration initiates a cascade of events, leading to the fusion of synaptic vesicles containing neurotransmitters with the presynaptic membrane. This fusion process, termed exocytosis, results in the release of neurotransmitters intothe synaptic cleft, a narrow gap separating the pre- and postsynaptic neurons. Released neurotransmitters then diffuse across the synaptic cleft and bind to specific receptors located on the postsynaptic membrane. These receptors can be broadly categorized as ionotropic or metabotropic. Ionotropic receptors, such as AMPA and NMDA receptors for glutamate, directly gate ion channels upon neurotransmitter binding, leading to rapid changes in membrane potential. For example, glutamate activation of AMPA receptors primarily elicits excitatory postsynaptic potentials (EPSPs) by allowing sodium ion influx. Conversely, GABA, a major inhibitory neurotransmitter, typically activates ionotropic receptors like GABAA, which primarily allow chloride ion influx, leading to inhibitory postsynaptic potentials (IPSPs) that hyperpolarize the postsynaptic neuron. Metabotropic receptors, on the other hand, indirectly influence neuronal activity through intracellular signaling cascades, often leading to slower but longer-lasting effects. This intricate interplay of neurotransmitter release, receptor activation, and ion channel gating forms the basis of synaptic transmission and enables the complex processing of information within the nervous system.
Neurons integrate multiple synaptic inputs through spatial and temporal summation. Spatial summation occurs when simultaneous inputs from different synapses combine, while temporal summation involves closely spaced inputs from the same synapse. The postsynaptic response depends on:
Synaptic strength, the efficacy of signal transmission at a synapse, is highly dynamic and undergoes activity-dependent modifications. Short-term plasticity encompasses transient changes in synaptic efficacy, typically lasting from seconds to minutes. These fluctuations are primarily driven by alterations in presynaptic calcium dynamics, impacting the release probability of neurotransmitters, and fluctuations in the availability of synaptic vesicles. In contrast, long-term plasticity, encompassing phenomena like long-term potentiation (LTP) and long-term depression (LTD), involves more enduring modifications in synaptic strength. These enduring changes are mediated by a diverse array of mechanisms, including: receptor trafficking, involving the insertion or removal of neurotransmitter receptors from the postsynaptic membrane; ?structural modifications at the synapse, such as the formation or elimination of dendritic spines; alterations in gene expression, leading to changes in the synthesis of proteins involved in synaptic function; and protein synthesis, resulting in the production of new proteins that contribute to the structural and functional plasticity of the synapse.
Neuromodulation encompasses a diverse array of physiological processes that subtly refine synaptic transmission within the nervous system. These processes operate through a variety of mechanisms, each contributing to the nuanced regulation of neural activity. One key mechanism involves the modulation of neurotransmitter release probability. Neuromodulators can influence the likelihood of neurotransmitter vesicles fusing with the presynaptic membrane, thereby altering the amount of neurotransmitter released into the synaptic cleft. This modulation can either enhance or diminish neurotransmission, depending on the specific neuromodulator and its target receptors. Furthermore, neuromodulators can exert significant influence on the sensitivity and responsiveness of postsynaptic receptors. By interacting with receptors or altering their molecular structure, neuromodulators can modify the strength of the postsynaptic response to a given neurotransmitter. This can lead to changes in the amplitude and duration of postsynaptic potentials, ultimately shaping the overall strength of synaptic transmission. Another critical aspect of neuromodulation lies in its ability to regulate the properties of ion channels. Neuromodulators can directly or indirectly influence the opening and closing of ion channels located on both pre- and postsynaptic neurons. This modulation of ion channel activity can alter the electrical excitability of neurons, thereby influencing their firing rate and overall contribution to neural circuits. Finally, neuromodulators can profoundly impact intracellular signaling pathways known as second messenger systems. These systems, triggered by the activation of specific receptors, can initiate a cascade of biochemical events within the neuron. Neuromodulators can modulate these second messenger systems, leading to changes in gene expression, protein synthesis, and long-term synaptic plasticity. Therefore, neuromodulation represents a complex and multifaceted phenomenon that plays a critical role in shaping the functional dynamics of neural circuits. By exerting a subtle yet profound influence on various aspects of synaptic transmission, neuromodulators contribute to the remarkable flexibility and adaptability of the nervous system.
This complex interplay of electrical and chemical signalling enables precise information processing in neural circuits, forming the basis for behavioral and cognitive functions. Understanding these mechanisms proves crucial for developing therapeutic strategies for neurological disorders affecting synaptic transmission [21-23].
4.2. Sensory and Motor Pathways
The nervous system orchestrates the body's ability to perceive, interpret, and respond to its environment through sensory and motor pathways. These pathways are integral to maintaining homeostasis, enabling voluntary movements, and reacting to external stimuli. Understanding their mechanisms and organization is crucial for comprehending how the nervous system facilitates seamless communication within the body.
4.2.1. Sensory Pathways
Sensory pathways are responsible for transmitting information from the external environment and internal physiological states to the central nervous system (CNS). These pathways rely on specialized sensory receptors and afferent neurons to detect and relay stimuli.
The nervous system relies on specialized cells known as sensory receptors to gather information about the internal and external environments. These receptors are classified based on the type of stimuli they detect. Mechanoreceptors respond to mechanical forces, such as pressure, vibration, and touch. Thermoreceptors detect changes in temperature, while nociceptors signal tissue damage and pain. Photoreceptors, primarily located in the retina, are responsible for vision by detecting light. Chemoreceptors respond to chemical stimuli, including changes in oxygen levels, taste, and smell. Upon activation, these receptors transduce the sensory information into electrical signals, specifically action potentials. These electrical signals are then transmitted along afferent neurons to the central nervous system (CNS) for further processing, ultimately leading to conscious perception or reflexive responses.
Sensory pathways, critical for





Arnab Roy*, Sonia Kumari, Aman Singh, Raj Kumar Gupta, Anuj Kumar, The Nervous System Decoded: Structural Dynamics, Functional Integration, and Emerging Neuroscientific Frontiers, Int. J. Sci. R. Tech., 2025, 2 (1), 28-52. https://doi.org/10.5281/zenodo.14594778
 10.5281/zenodo.14594778
10.5281/zenodo.14594778