
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
1Ph.D. Scholar (Botany), Kalinga University, Kotni, Atal Nagar-Nava Raipur, Chhattisgarh 492101, India.?
2Ph.D. Scholar (Biotechnology), Kalinga University, Kotni, Atal Nagar-Nava Raipur, Chhattisgarh 492101, India.
3Ph.D. Scholar (Pharmacy), Kalinga University, Kotni, Atal Nagar-Nava Raipur, Chhattisgarh 492101, India.
4Student, B. Pharm, Kalinga University, Kotni, Atal Nagar-Nava Raipur, Chhattisgarh 492101, India
The dwarf water clover (Marsilea minuta), a versatile aquatic fern, has long been recognized in traditional medicine for its wide-ranging therapeutic applications. This review provides a comprehensive exploration of the botanical, phytochemical, and pharmacological attributes of M. minuta, emphasizing its potential as a source of novel medicinal agents. Botanically, M. minuta is characterized by its distinct four-lobed leaf structure and adaptability to various aquatic and terrestrial environments, making it a sustainable resource for therapeutic exploitation. Phytochemical investigations have revealed the presence of diverse bioactive compounds, including flavonoids, phenolic acids, alkaloids, and saponins, which are associated with its antioxidant, anti-inflammatory, anxiolytic, and hypoglycemic activities. Pharmacological studies underscore its role in modulating key biological pathways, such as neurotransmitter regulation for anxiolytic effects and enzymatic inhibition in carbohydrate metabolism for antidiabetic applications. Preclinical evidence supports its efficacy in treating conditions like anxiety, epilepsy, fever, and inflammation. Additionally, its antioxidant properties suggest a protective role against oxidative stress-related disorders. However, the therapeutic potential of M. minuta is tempered by the limited availability of toxicological and clinical studies, highlighting the need for comprehensive safety evaluations and standardized dosing guidelines. This review also identifies critical gaps in current research, including the need for molecular mechanism elucidation, advanced pharmacokinetic profiling, and well-designed clinical trials. By integrating traditional knowledge with modern pharmacological approaches, M. minuta can be harnessed for its full therapeutic potential. The findings presented in this review aim to serve as a foundation for future studies, paving the way for the development of evidence-based applications of M. minuta in contemporary medicine.
Marsilea minuta, commonly known as dwarf water clover or water shamrock, represents a fascinating aquatic pteridophyte within the family Marsileaceae, demonstrating remarkable adaptability across diverse aquatic ecosystems [1, 2]. This diminutive heterosporous fern exhibits a distinctive creeping rhizome system that facilitates its spread across wetland substrates, while its characteristic quadrifoliate fronds float delicately on water surfaces or stand erect in shallow conditions [3, 4]. The species has evolved specialized anatomical features, including aerenchymatous tissues and modified stomatal complexes, enabling efficient gas exchange and survival in both submerged and emergent conditions [5, 6]. Its reproductive biology is particularly intriguing, featuring sporocarps that demonstrate exceptional resilience, capable of remaining viable for extended periods under adverse conditions, thus ensuring species perpetuation across seasonal wetlands [7, 8].
? ? ? ?
? ? ? ? ? ? 
? ? ? ? ? ??
Fig. 1. Dwarf Water Clover (Marsilea minuta)
In the rich tapestry of traditional medicine, M. minuta has established itself as a cornerstone therapeutic agent, particularly within the indigenous healthcare systems of Southeast Asian countries like Thailand, Vietnam, and Indonesia, as well as South Asian countries like Bangladesh, India, and Sri Lanka [9, 10, 11]. Local healers have long recognized its medicinal value, incorporating the plant into various therapeutic preparations for treating ailments ranging from digestive disorders to inflammatory conditions [12, 13]. The ethnomedicinal applications of this species reflect sophisticated traditional knowledge systems developed through generations of empirical observation and practical application [14].
The scientific community's interest in M. minuta has experienced a significant surge in recent decades, driven by the growing recognition of its potential therapeutic applications and the global shift toward evidence-based validation of traditional medicines [15, 16]. Modern research initiatives have begun unraveling the complex phytochemical profile of this species, revealing a diverse array of bioactive compounds including flavonoids, terpenoids, alkaloids, and phenolic compounds [17, 18, 19]. These investigations have expanded beyond mere chemical characterization to encompass detailed pharmacological studies, exploring mechanisms of action and potential therapeutic applications [20]. The emerging scientific evidence suggests promising activities including antidiabetic, anti-inflammatory, antioxidant, and antimicrobial properties, warranting further investigation for potential drug development [21, 22, 23].
Contemporary research approaches, utilizing advanced analytical techniques and molecular biology tools, have initiated a new era in understanding M. minuta's therapeutic potential [24, 25]. This scientific renaissance has been characterized by systematic investigations into its bioactive constituents, safety profiles, and mechanism of action at the cellular and molecular levels [26]. The convergence of traditional knowledge and modern scientific methodology has created a robust framework for developing novel therapeutic agents from this historically significant plant [27, 28]. This paradigm shift from empirical usage to evidence-based application represents a critical evolution in the understanding and utilization of M. minuta's medicinal properties, potentially paving the way for innovative pharmaceutical developments while simultaneously validating centuries of traditional medicinal knowledge [29, 30].
The root system of Marsilea minuta exhibits a complex and highly adaptive architectural organization, characterized by an extensive rhizomatous network that serves multiple physiological and structural functions [31]. The primary roots emerge systematically from specialized nodal regions along the creeping rhizome, displaying positive geotropism and forming the foundational framework of the plant's anchorage system [32, 33].?
? ? ? ?
? ? ? ? ? ? 
? ? ? ? ? ??
Fig. 2. TS of M. minuta Root
These primary roots, measuring approximately 0.2-0.5 mm in diameter, demonstrate remarkable plasticity in their growth patterns and morphological characteristics depending on environmental conditions [34]. They maintain a consistent circadian rhythm in their growth rate, with maximum elongation typically occurring during nocturnal periods. The development of adventitious roots represents a significant adaptive feature of M. minuta, occurring at regular intervals of 2-4 cm along the rhizome axis. These adventitious roots demonstrate remarkable physiological adaptability, possessing the capacity to modify their anatomical structure based on environmental conditions [35, 36]. In terrestrial environments, they exhibit enhanced vascular tissue development and increased aerenchyma formation, while in aquatic conditions, they show modified cortical tissue arrangements optimized for nutrient absorption from water [37, 38].
The secondary root system comprises an intricate network of fine, dichotomously branching roots that form dense, mat-like structures [39]. These secondary roots display sophisticated branching patterns governed by both endogenous hormonal controls and environmental stimuli [40]. Their development follows a precise temporal and spatial organization, with lateral root primordia forming in the pericycle region of the primary roots. The branching pattern typically exhibits a hierarchical organization, with secondary roots further subdividing into tertiary branches, creating an efficient soil exploration network [41, 42].
Root hair development in M. minuta demonstrates remarkable environmental plasticity, with abundant production in terrestrial forms where they play crucial roles in water and nutrient absorption, soil adherence, and rhizosphere interactions [43, 44]. These root hairs typically measure 0.5-1.5 mm in length and exhibit a density of approximately 200-300 hairs per square millimeter of root surface. Conversely, in aquatic forms, root hair development is significantly reduced, reflecting the altered requirements for nutrient and water absorption in aquatic environments [45]. This morphological plasticity is regulated by complex interactions between environmental signals and endogenous hormonal pathways, particularly involving auxin and ethylene signaling networks [46, 47].
The entire root system demonstrates remarkable physiological integration, with sophisticated hormone-mediated communication networks coordinating growth and development across different root types [48]. This integration enables efficient resource allocation and optimal adaptation to varying environmental conditions, whether terrestrial or aquatic. The root system also exhibits significant symbiotic associations with beneficial soil microorganisms, including arbuscular mycorrhizal fungi, which enhance the plant's nutrient acquisition capabilities and stress tolerance mechanisms [49, 50].
The rhizome system of Marsilea minuta displays a complex and well-adapted morphological architecture that significantly contributes to its survival and propagation in diverse aquatic and semi-aquatic environments [51]. The creeping rhizome exhibits a pronounced horizontal growth pattern, characterized by internodes measuring 1-3 centimeters in length, which allows for efficient substrate colonization and resource acquisition [52]. This growth pattern is particularly advantageous in shallow water bodies and muddy substrates, where the rhizome can effectively anchor the plant while exploring new territories for nutrient acquisition. The dichotomous branching pattern of the rhizome represents a sophisticated evolutionary adaptation, facilitating extensive vegetative propagation and enabling the plant to establish dense populations in favorable habitats [53, 54].
? ? ? ?
? ? ? ? ? ? 
? ? ? ? ? ??
Fig. 3 TS of M. minuta Rhizome
?This branching mechanism involves the formation of two equal branches from a single growing point, with each branch capable of independent growth and further dichotomous divisions. The rhizome's external appearance undergoes significant ontogenetic changes, transitioning from a light brown coloration in younger segments to a distinctive dark brown or black pigmentation in mature portions, ?primarily due to the accumulation of phenolic compounds and structural modifications in the epidermal layer [55, 56, 57]. The internal anatomy reveals a highly specialized vascular system comprising a modified protostele, which represents an evolutionary advancement in pteridophytes. This vascular arrangement consists of a central xylem core surrounded by phloem tissue, with endodermal and pericycle layers providing additional structural and physiological support [58]. The storage tissue system within the rhizome is remarkably developed, containing abundant starch grains arranged in specialized amyloplasts, along with other essential nutrients including proteins and lipids [60]. These storage tissues play crucial roles in the plant's survival during unfavourable environmental conditions and support rapid growth during favourable periods. The rhizome also contains specialized secretory cells that produce various bioactive compounds, including flavonoids and terpenoids, which contribute to the plant's chemical defense mechanisms and medicinal properties [61]. The epidermal layer of the rhizome is fortified with a thick cuticle and produces root hairs at regular intervals, enhancing water and nutrient absorption while also providing additional anchorage. This sophisticated rhizome structure enables M. minuta to adapt to fluctuating water levels and varying substrate conditions, making it a successful colonizer in diverse aquatic ecosystems. Furthermore, the rhizome's architectural features facilitate efficient nutrient translocation and resource sharing between connected ramets, contributing to the plant's clonal growth strategy and ecological success in its natural habitats [62, 63].
? ? ? ?
? ? ? ? ? ? 
? ? ? ? ?
? ??Fig. 4 TS of M. minuta Petiole
The leaves of Marsilea minuta exhibit exceptional heterophylly, showcasing remarkable adaptive plasticity in response to varying environmental conditions [64]. Each compound leaf manifests as a distinctive quadrifoliate structure, bearing four symmetrically arranged leaflets that are reminiscent of a four-leaf clover. These leaflets demonstrate significant morphological variations depending on their growth environment, with dimensions typically ranging from 0.5 to 1.2 cm in length [65]. The leaflets display an obdeltoid to cuneate shape, with the broader end positioned distally and gradually tapering towards the attachment point. The petioles exhibit remarkable environmental adaptability, varying dramatically in length from 3 to 15 centimeters, with the specific length correlating directly with water depth in aquatic environments. This adaptive mechanism ensures optimal leaf positioning for photosynthetic efficiency regardless of water level fluctuations [66, 67].
? ? ? ?
? ? ? ? ? ? 
? ? ? ?
Fig. 4 TS of M. minuta Petiole
The venation pattern within each leaflet is particularly noteworthy, characterized by repeated dichotomous branching that creates an intricate network of vascular tissues. This specialized venation architecture not only provides structural support but also ensures efficient nutrient and water distribution throughout the leaf tissue. A fascinating aspect of M. minuta's leaf anatomy is its stomatal distribution and density, which varies significantly between terrestrial and aquatic forms [68, 69]. In terrestrial morphs, stomata are abundantly present on both adaxial and abaxial leaf surfaces, facilitating efficient gas exchange in the atmospheric environment. However, aquatic forms exhibit a marked reduction in stomatal density, particularly on the abaxial surface, representing an evolutionary adaptation to submerged conditions where gas exchange primarily occurs through direct diffusion across the leaf surface [70, 71]. The epidermal cells in aquatic forms also tend to be thinner and more elongated compared to their terrestrial counterparts, enhancing the plant's ability to absorb nutrients directly from the aqueous environment. Furthermore, the leaflets demonstrate significant modifications in their internal anatomy, with aquatic forms showing reduced development of palisade mesophyll and increased aerenchyma tissue, while terrestrial forms maintain a more typical dorsiventral leaf structure with well-defined palisade and spongy mesophyll layers [72]. This remarkable morphological plasticity in leaf structure represents a sophisticated evolutionary adaptation that enables M. minuta to thrive in both terrestrial and aquatic environments, making it an excellent model organism for studying plant adaptation mechanisms [73, 74].
The reproductive structures of Marsilea minuta exhibit a remarkably sophisticated morphological organization, characterized by distinctive sporocarps that serve as the primary vessels for sexual reproduction [75]. These specialized reproductive organs manifest as bean-shaped structures, typically arising from abbreviated peduncles that emerge near the bases of petioles or along the rhizome. The sporocarps demonstrate bilateral symmetry and possess a remarkably resilient outer wall composed of multiple sclerenchymatous layers, presenting a dark brown to almost black coloration at maturity [76, 77].
? ? ? ?
? ? ? ? ? ? 
? ? ? ? ? ??
Fig. 5. LS of M. minuta Sporocarp
This protective exterior exhibits intricate ridge patterns that form species-specific geometric configurations, often serving as crucial taxonomic markers for species identification within the Marsileaceae family [78]. The sporocarp wall's complex ultrastructure consists of multiple layers: an outer epidermis with thick cuticular deposits, a subepidermal prismatic layer, and several inner layers of sclerenchymatous cells arranged in distinctive patterns that contribute to the sporocarp's mechanical strength and water impermeability [79]. Internally, the sporocarp demonstrates a sophisticated compartmentalization system, with multiple sori arranged in two rows along a specialized tissue called the sorophore [80]. Each sorus contains both megasporangia and microsporangia, representing an advanced level of heterosporous reproduction among pteridophytes [81]. The megasporangia, typically larger and fewer in number, produce single megaspores, while the more numerous microsporangia generate multiple microspores. This dimorphic spore production system represents an evolutionary advancement in reproductive efficiency. The sporocarps typically measure between 2-4 millimeters in length, though size variations can occur based on environmental conditions and geographical location. The external surface ornamentation patterns involve a complex network of raised ridges, tubercles, and sometimes minute depressions that form specific geometric patterns [82, 83]. These surface features play crucial roles in water absorption during germination and may influence spore dispersal mechanisms. The sporocarp's internal architecture includes a gelatinous ring structure that swells upon hydration, creating mechanical pressure that facilitates sporocarp dehiscence and subsequent spore release. This sophisticated reproductive system allows M. minuta to maintain genetic diversity while ensuring successful propagation in both aquatic and terrestrial environments. The sporocarp's structural adaptations, particularly its hard outer wall and internal compartmentalization, enable long-term spore viability and protection against environmental stressors, contributing to the species' successful colonization of diverse habitats across tropical and subtropical regions [84, 85].
Marsilea minuta exhibits remarkable heterosporous reproduction, characterized by the production of two distinct types of spores that showcase sophisticated adaptations for survival and reproduction in aquatic environments [86]. The microspores, which develop within microsporangia, are spherical structures measuring 25-35 ?m in diameter and are produced in substantial quantities, often exceeding 50,000 per sporocarp. These microspores possess a complex wall architecture comprising three distinct layers: the outermost perispore (characterized by its sculptured surface), the middle exospore (composed of sporopollenin), and the innermost endospore (primarily cellulosic in nature). This trilayered structure provides essential protection against environmental stressors while facilitating the eventual release of male gametophytes [87, 88].
In contrast, the megaspores, developed within megasporangia, are significantly larger, oval-shaped structures measuring 400-600 ?m in length. Each megaspore demonstrates remarkable organizational complexity, featuring a prominent apical region where archegonia later develop. The megaspore wall exhibits a more elaborate stratification compared to microspores, with specialized layers including an outer epispore (featuring distinctive surface ornamentation), a robust exospore (containing high concentrations of sporopollenin and silica deposits), and an inner endospore layer rich in polysaccharides [90, 91]. This intricate wall structure not only provides mechanical protection but also facilitates specific physiological processes essential for germination [92].
The spore germination process in M. minuta is particularly fascinating, showing sophisticated adaptation to its aquatic habitat. Upon hydration, both spore types undergo carefully orchestrated developmental sequences [93]. The microspores initiate rapid germination within 6-12 hours of hydration, releasing small, reduced male gametophytes that produce motile spermatozoids [94]. The megaspores, however, demonstrate a more complex germination pattern, requiring 12-24 hours of hydration before initiating development. During germination, the megaspore undergoes controlled wall rupture at the apical region, followed by the emergence of a female gametophyte bearing archegonia. This temporal separation in germination timing represents an evolutionary adaptation that optimizes reproductive success by ensuring the mature development of female gametophytes before the release of male gametes [95, 96].
The entire reproductive cycle is further enhanced by various structural and physiological adaptations. The spore walls contain specific compounds, including phenolics and sporopollenin, which provide protection against UV radiation and microbial degradation [97]. Additionally, the spore surfaces feature specialized structures that facilitate water absorption and regulate gas exchange, crucial for successful germination. The presence of lipid bodies and starch reserves within the spores ensures sufficient energy resources for the developing gametophytes, while specialized proteins and enzymes facilitate the controlled breakdown of wall materials during germination [98, 99].
This sophisticated reproductive system, characterized by distinct spore types with specialized structures and germination mechanisms, exemplifies the evolutionary adaptations that have enabled M. minuta to successfully colonize and reproduce in various aquatic environments, contributing to its widespread distribution across tropical and subtropical regions [100, 101].
The anatomical investigation of Marsilea minuta reveals a complex and well-organized internal structure characteristic of advanced pteridophytes. The outermost layer comprises a distinctly organized epidermis fortified with a substantial cuticle layer, which serves multiple functions including mechanical protection and regulation of water loss through transpiration [102]. This cuticular layer demonstrates significant thickness variation between terrestrial and submerged forms, exhibiting adaptive plasticity to different environmental conditions. The epidermal cells are arranged in a compact, continuous layer with specialized guard cells forming stomata, whose frequency notably varies between the adaxial and abaxial surfaces [103].
? ? ? ?
? ? ? ? ? ? 
? ? ? ? ? ??
Fig. 6. TS of M. minuta Stem
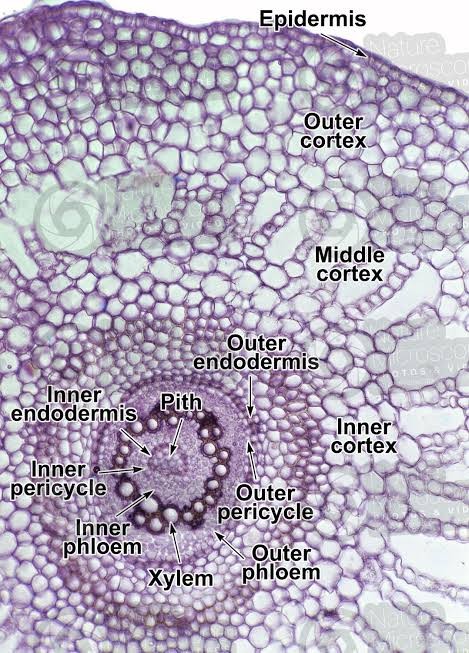
Beneath the epidermis lies a remarkably differentiated cortex, which is clearly demarcated into outer and inner regions. The outer cortical region consists of 3-5 layers of collenchymatous cells with pronounced corner thickenings, providing mechanical support while maintaining tissue flexibility. In contrast, the inner cortical region exhibits large, thin-walled parenchymatous cells arranged with conspicuous intercellular spaces forming aerenchyma, particularly well-developed in submerged portions of the plant [104, 105]. This adaptation facilitates efficient gas exchange and provides buoyancy in aquatic environments. A well-defined endodermis with prominent Casparian strips separates the cortex from the central stele, playing a crucial role in selective nutrient transport and water regulation. The Casparian strips, composed of suberin and lignin deposits in the radial and transverse walls, create an efficient barrier controlling the movement of substances between the cortex and stele [106]. The central stele demonstrates a sophisticated organization with xylem elements arranged in a characteristic V-pattern, a feature distinguishing M. minuta from other species within the genus. The metaxylem vessels, larger in diameter compared to the protoxylem, are positioned towards the arms of the V, while smaller protoxylem elements are located at the base, indicating an exarch maturation pattern [107]. The phloem tissue is strategically distributed in a peripheral pattern around the xylem, consisting of sieve cells and companion cells, optimizing the transport of photosynthates throughout the plant body. This vascular arrangement maximizes the efficiency of water and nutrient transport while maintaining structural integrity. Between the xylem and phloem, a layer of cambiform cells facilitates limited secondary growth, an unusual feature among pteridophytes [108, 109]. The entire stele is encased in a distinct pericycle, composed of one to two layers of parenchymatous cells, which maintains the potential for lateral root initiation. This intricate anatomical organization reflects the evolutionary adaptations of M. minuta to its amphibious lifestyle, enabling efficient resource allocation and structural support in both aquatic and terrestrial environments.
The leaf structure of Marsilea minuta exhibits remarkable aquatic adaptations that showcase the species' evolutionary success in both aquatic and semi-aquatic environments. The most notable adaptation is the significant reduction in cuticle thickness, particularly in submerged leaves, which facilitates enhanced gas exchange and nutrient absorption directly from the surrounding water [110, 111]. This reduced cuticle, ranging from 0.5-2.0 ?m in thickness, is complemented by a modified epidermal layer featuring specialized cells that optimize the plant's interaction with its aquatic medium. The development of aerenchyma tissue represents another crucial adaptation, forming an intricate network of interconnected air channels throughout the leaf tissue. These aerenchymatous spaces, which can occupy up to 40% of the leaf volume, not only provide buoyancy but also ensure efficient gas exchange and oxygen transport to submerged plant parts, enabling survival in oxygen-depleted aquatic environments. The chlorenchyma arrangement in M. minuta leaves demonstrates remarkable plasticity, with palisade cells showing variable arrangements depending on the degree of submergence [112, 113].
? ? ? ?
? ? ? ? ? ? 
? ? ? ? ? ??
Fig. 7. TS of M. minuta leaf
In fully submerged leaves, the palisade tissue becomes more compact and exhibits a modified orientation to maximize light capture under water, while maintaining sufficient intercellular spaces for gas exchange. The presence of specialized mechanical tissues at leaflet joints represents an intricate adaptation that allows the four leaflets to respond to water movement and light conditions. These joints contain collenchymatous and sclerenchymatous tissues arranged in a distinctive pattern, enabling the leaflets to fold together during unfavorable conditions or at night, and to spread horizontally when environmental conditions are optimal. The vascular bundle patterns in M. minuta leaves show unique modifications that differ significantly from terrestrial ferns. The primary vein divides dichotomously within each leaflet, forming a complex network of smaller veins with enhanced xylem elements for water transport and modified phloem tissue for efficient nutrient translocation [114, 115]. The bundle sheath cells surrounding these vascular tissues are notably enlarged and contain numerous chloroplasts, contributing to the plant's photosynthetic efficiency in aquatic conditions. Additionally, the presence of hydropoten cells in the epidermal layer facilitates selective ion absorption and secretion, maintaining optimal osmotic balance in varying water conditions [116]. These structural modifications collectively demonstrate the sophisticated level of adaptation that M. minuta has evolved to thrive in its aquatic habitat while maintaining the flexibility to survive in temporarily dry conditions, making it an excellent example of evolutionary adaptation to amphibious environments.
Marsilea minuta, commonly known as water clover, has a unique and intricate life cycle that reflects its adaptability and biological complexity. The life cycle can be broadly categorized into distinct developmental stages, each characterized by specific biological processes and time frames, influenced by environmental conditions such as temperature, light, and moisture availability [117, 118]. The life cycle begins with the spore germination phase, a critical initial stage lasting approximately 3 to 5 days under optimal conditions. Spores, which serve as the reproductive units of this fern, are typically dispersed in aquatic or moist environments. Upon encountering favorable conditions, these spores undergo germination [119]. The process involves the absorption of water, metabolic activation, and the emergence of the prothallus, a small, flat, green structure that serves as the gametophyte. Successfulgermination is influenced by factors such as pH, temperature, and the availability of nutrients, which collectively dictate the viability and growth rate of the gametophyte [120].
? ? ? ?
? ? ? ? ? ? 
? ? ? ? ? ??
Fig. 8. Developmental stage of M. minuta Microsporangium and Megasporangium
Following spore germination, the gametophyte development phase occurs over 10 to 15 days. During this stage, the prothallus matures and develops reproductive structures known as antheridia and archegonia, which produce male and female gametes, respectively. This phase is crucial for sexual reproduction, as the motile sperm cells require a thin film of water to swim toward the archegonia to fertilize the egg cells [121, 122]. This dependence on water highlights the evolutionary significance of Marsilea minuta's aquatic or semi-aquatic habitats. Successful fertilization marks the transition to the next stage of development. The fertilized egg, or zygote, then progresses into the young sporophyte establishment phase, which spans approximately 20 to 30 days. During this stage, the sporophyte, representing the diploid phase of the life cycle, begins to emerge from the gametophyte [123, 124]. This young sporophyte is initially reliant on the gametophyte for nutrients but gradually develops its independent structures, such as roots, stems, and fronds. These structures enable the plant to anchor itself in the substrate and initiate photosynthesis, ensuring its survival and growth. The successful establishment of a young sporophyte depends heavily on environmental conditions, particularly light intensity and water quality [125].
The final phase, the mature plant development phase, takes around 45 to 60 days. During this stage, the sporophyte achieves full maturity, characterized by the production of well-developed leaves and reproductive structures known as sporocarps. These sporocarps produce and release spores, thereby completing the life cycle and facilitating the continuation of the species. The mature plant also plays a vital ecological role by contributing to the aquatic ecosystem, serving as a habitat for microorganisms, and influencing nutrient cycling [126, 127].
Marsilea minuta, exhibits remarkable adaptability to its environment. This adaptability is driven by its ability to undergo physiological and morphological changes in response to varying environmental conditions [128]. These adaptive strategies enable the plant to thrive across diverse habitats, making it a versatile species with considerable ecological significance.
One notable adaptation of M. minuta is its ability to rapidly elongate its petiole in response to changes in water levels. This characteristic allows the plant to maintain optimal positioning of its leaves for photosynthesis and gas exchange, irrespective of fluctuating water depths. By extending the petiole, the plant ensures that its photosynthetic apparatus remains above water, maximizing exposure to sunlight [129, 130]. This dynamic adjustment in growth is regulated by hormonal changes, particularly the involvement of auxins and gibberellins, which promote cell elongation in response to submergence or water scarcity. Another significant adaptation is the diurnal movement of its leaves. The leaflets of M. minuta exhibit nyctinastic movements, unfolding during daylight to capture sunlight for photosynthesis and folding at night to minimize water loss and reduce exposure to nocturnal predators or adverse environmental factors [131]. These movements are governed by changes in turgor pressure within specialized cells at the base of the leaflets, known as pulvini. This adaptation highlights the plant's efficient energy conservation mechanisms and its ability to synchronize physiological processes with external environmental cues [132].
The plant also demonstrates a remarkable ability to modify its leaf structure under different light intensities. In high light conditions, M. minuta develops thicker, more robust leaves with increased pigmentation to prevent photodamage and optimize photosynthetic efficiency [133]. Conversely, under low light conditions, the plant produces thinner leaves with larger surface areas to maximize light capture. These structural modifications underline the plasticity of the plant?s photosynthetic machinery and its ability to adapt to varying light environments [134].
Furthermore, M. minuta exhibits altered reproductive timing in response to environmental cues, ensuring its survival and reproductive success. The plant can modulate its spore production based on factors such as water availability, temperature fluctuations, and seasonal changes [135]. For instance, in favorable conditions, the plant prioritizes vegetative growth and reproduction, while during adverse conditions, it enters a dormant phase, conserving resources and ensuring the survival of reproductive structures like spores. These adjustments underscore the plant?s capacity to anticipate and respond to environmental challenges, ensuring long-term population stability [136, 137].
In summary, the adaptive strategies of M. minuta reflect a finely tuned interplay between morphological, physiological, and reproductive mechanisms. By elongating its petiole in response to water level changes, displaying diurnal leaf movements, modifying leaf structure under varying light intensities, and altering reproductive timing based on environmental cues, M. minuta exemplifies the resilience and versatility of aquatic ferns [138, 139]. Understanding these adaptations not only highlights the ecological significance of this plant but also provides insights into its potential applications in ecological restoration and aquaculture systems.
Marsilea minuta, also known for its diverse pharmacological properties, the plant has garnered significant attention in the field of phytochemistry. The phytochemical constituents of M. minuta are the primary drivers of its therapeutic potential, making it a subject of interest for researchers in natural product chemistry.
Flavonoids are a significant class of phytochemicals found in Marsilea minuta, contributing extensively to its medicinal properties. These naturally occurring compounds are renowned for their potent antioxidant activity, which is essential in neutralizing free radicals and mitigating oxidative stress in biological systems. Oxidative stress is implicated in the pathogenesis of various chronic diseases, including cardiovascular disorders, neurodegenerative diseases, and cancer. By scavenging reactive oxygen species (ROS), flavonoids play a critical role in protecting cells from oxidative damage [140, 141].
Research has identified the presence of flavonoids like quercetin and kaempferol derivatives in M. minuta, both of which are well-studied for their diverse biological activities. Quercetin, a flavonol, is known for its strong antioxidant capacity, anti-inflammatory action, and ability to modulate immune responses [142]. Similarly, kaempferol, another flavonol present in M. minuta, exhibits similar antioxidant potential, along with significant anti-inflammatory and antimicrobial properties. These flavonoid compounds contribute to the plant?s pharmacological profile, enhancing its therapeutic value [143].
The anti-inflammatory properties of flavonoids in M. minuta are particularly noteworthy. Inflammation is a key factor in many chronic conditions, and flavonoids can modulate inflammatory pathways by inhibiting the production of pro-inflammatory cytokines and enzymes such as cyclooxygenase (COX) and lipoxygenase (LOX) [144, 145]. This inhibition reduces the release of inflammatory mediators, thereby alleviating symptoms of inflammation. As a result, M. minuta has the potential to be utilized in managing conditions like arthritis, inflammatory bowel disease, and other inflammation-related disorders [146].
Furthermore, the antimicrobial activities of flavonoids present in M. minuta are significant, as these compounds can disrupt the growth of various pathogenic microorganisms. By targeting bacterial cell walls, altering membrane permeability, and interfering with microbial enzyme activity, flavonoids in M. minuta exhibit broad-spectrum antimicrobial effects [147]. This makes the plant a potential candidate for developing natural antimicrobial agents, especially in an era where antibiotic resistance is a growing global concern.
In addition to its antioxidant and antimicrobial actions, M. minuta 's flavonoids also possess neuroprotective properties. These compounds protect neural cells from oxidative damage, a leading cause of neurodegenerative diseases such as Alzheimer's and Parkinson's disease [148, 149]. By reducing oxidative stress and inflammation in the nervous system, flavonoids contribute to maintaining cognitive function and preventing neurodegeneration. Furthermore, flavonoids have been shown to enhance neuronal survival and promote synaptic plasticity, which are critical for learning and memory processes [150].
Phenolic compounds represent a major group of phytochemicals present in Marsilea minuta, known for their powerful antioxidant properties and their capacity to neutralize reactive oxygen species (ROS) [151]. These compounds play a critical role in protecting cells from oxidative damage, which is often linked to the development of chronic diseases such as cancer, diabetes, and cardiovascular disorders. The antioxidant activity of phenolic compounds helps in reducing oxidative stress, thereby preventing the harmful effects of free radicals that can lead to cellular and tissue damage [152, 153].
Among the phenolics identified in M. minuta, gallic acid and caffeic acid are of particular interest due to their wide-ranging pharmacological properties. Gallic acid, a trihydroxybenzoic acid, is renowned for its strong antioxidant, anti-inflammatory, and antimicrobial activities [154, 155]. It acts by scavenging free radicals and inhibiting lipid peroxidation, processes that are essential in preventing the onset of oxidative stress-related disorders. Additionally, gallic acid has been shown to modulate enzymatic pathways that are involved in inflammation, contributing to its anti-inflammatory effects, which are valuable in the treatment of inflammatory diseases [156].
Caffeic acid, another phenolic acid found in M. minuta, is similarly noted for its antioxidant properties, as well as its anti-inflammatory, antidiabetic, and anticancer effects. Caffeic acid works by inhibiting ROS production and enhancing the body's antioxidant defense mechanisms, including the upregulation of endogenous antioxidant enzymes such as superoxide dismutase (SOD) and catalase [157, 158]. This phenolic compound also exhibits anti-hyperglycemic effects, which are beneficial in managing diabetes. Studies have shown that caffeic acid can improve insulin sensitivity and glucose metabolism, reducing the risk of complications associated with diabetes, such as cardiovascular diseases.
The antioxidant capacity of phenolic compounds like gallic acid and caffeic acid not only protects against oxidative stress but also supports vascular health by preventing the oxidation of low-density lipoprotein (LDL) cholesterol, a key factor in the development of atherosclerosis [159]. By inhibiting the oxidative modification of LDL, these compounds reduce the risk of plaque formation in blood vessels, which can lead to cardiovascular events such as heart attacks and strokes. Furthermore, the anti-inflammatory effects of these phenolics contribute to the stabilization of vascular endothelium, thereby improving overall cardiovascular function [160].
The therapeutic potential of M. minuta in managing chronic diseases like diabetes and cardiovascular disorders is largely attributed to its rich content of phenolic compounds. By mitigating oxidative damage, reducing inflammation, and enhancing metabolic function, these compounds provide a multi-faceted approach to disease prevention and management. The presence of gallic acid, caffeic acid, and other phenolic compounds in M. minuta highlights the plant's value as a natural source of bioactive molecules with significant health-promoting properties [161, 162]. Continued research into the phenolic profile of M. minuta may further elucidate its potential in the development of novel therapeutic agents for a range of oxidative stress-related conditions.
Tannins, a diverse class of phenolic compounds, are present in significant quantities in Marsilea minuta, playing a crucial role in its pharmacological properties. Known for their strong astringent qualities, tannins contribute to the plant?s ability to aid in wound healing by contracting tissues and reducing inflammation [163]. This property is especially beneficial in closing wounds, preventing excessive bleeding, and promoting the formation of new tissue. Additionally, tannins have been well-documented for their potent antimicrobial activities, making them effective in inhibiting the growth of various bacteria, fungi, and other pathogens that can cause infections. Their antimicrobial action is primarily due to their ability to bind with microbial proteins and enzymes, thereby disrupting cellular processes essential for microbial survival [164, 165].
The high tannin content in M. minuta plays a pivotal role in its inclusion in traditional medicinal practices, where it is frequently used for treating infections and skin ailments. In traditional settings, extracts or preparations made from the plant are applied topically to treat wounds, cuts, and ulcers, taking advantage of the tannins' ability to create a protective barrier over the affected area [166]. This helps in minimizing contamination and accelerating the healing process. Moreover, tannins are known to have anti-inflammatory properties, which further enhance the plant's therapeutic value in managing skin conditions, inflammation, and microbial infections [167].
Beyond their wound-healing and antimicrobial properties, tannins also exhibit antioxidant activity, which contributes to their overall therapeutic benefits. By neutralizing free radicals and reducing oxidative stress, tannins support the body?s defense mechanisms against cellular damage, particularly in cases where infection and tissue damage are involved. This antioxidant capability adds another dimension to the medicinal use of M. minuta, as it suggests potential protective effects on a cellular level, contributing not only to faster recovery from wounds but also to the prevention of further tissue damage during infection or injury [168, 169].
In addition to topical applications, the internal use of tannin-rich extracts from M. minuta in traditional medicine highlights their broader systemic effects. The astringent properties of tannins are known to aid in managing conditions like diarrhea and gastrointestinal infections by reducing excessive fluid secretion and tightening mucosal tissues [170]. This reflects the versatility of tannins in addressing both external and internal health issues.
Overall, the abundance of tannins in M. minuta significantly enhances its medicinal value, supporting its use in both modern and traditional medicine. These compounds not only contribute to wound healing and infection control but also exhibit a wide range of bioactivities, including antioxidant, anti-inflammatory, and astringent effects. The continued exploration of tannins from M. minuta may provide further insights into their therapeutic potential, expanding their applications in pharmacology and natural medicine [171].
Alkaloids represent a complex class of naturally occurring organic compounds characterized by the presence of one or more nitrogen atoms within heterocyclic ring structures. In Marsilea minuta, while alkaloid concentrations are relatively modest compared to other bioactive constituents, chromatographic analyses have revealed the presence of specific alkaloid derivatives that contribute to the species' pharmacological profile [172, 173]. These nitrogenous compounds demonstrate noteworthy biological activities, particularly in modulating nociceptive pathways and inflammatory cascades. Mechanistically, certain alkaloids isolated from M. minuta have been shown to inhibit prostaglandin synthesis and influence cytokine production, thereby contributing to the plant's analgesic and anti-inflammatory properties [174, 175]. The structural diversity of these alkaloids, including variations in their ring systems and substitution patterns, likely underlies their distinct molecular interactions with biological targets. Recent phytochemical investigations suggest these compounds may act synergistically with other constituent molecules, though further research is needed to fully elucidate their structure-activity relationships and precise mechanisms of action in therapeutic applications.
Saponins, glycosidic compounds characterized by their amphipathic molecular structure, represent a complex class of secondary metabolites isolated from Marselia minuta with multifaceted biological significance. These compounds demonstrate remarkable biochemical versatility through their intrinsic surfactant-like properties, which facilitate diverse physiological interactions at molecular and cellular levels [176, 177]. Biochemical investigations reveal their capacity to modulate lipid metabolism by interfering with cholesterol absorption and transportation mechanisms, thereby potentially contributing to cardiovascular homeostasis. Immunomodulatory characteristics of saponins are evidenced through their ability to stimulate immune cell proliferation and cytokine production, suggesting potential therapeutic implications in immunological regulation [178]. The antimicrobial efficacy of these compounds stems from their structural configuration, enabling membrane disruption and inhibition of microbial growth through surfactant-mediated mechanisms. Moreover, their anti-inflammatory properties are attributed to complex molecular interactions involving signaling pathways and inflammatory mediator suppression. The intricate chemical structure of saponins, comprising hydrophilic glycoside moieties and hydrophobic aglycone regions, enables sophisticated biomembrane interactions, underlining their significant pharmaceutical and nutraceutical potential in contemporary biomedical research [179].
The terpenoids found in Marsilea minuta play a significant role in the plant's aromatic characteristics and therapeutic potential. These natural compounds, which encompass monoterpenes and diterpenes, are widely recognized for their diverse biological activities. Notably, they have demonstrated anti-inflammatory, antimicrobial, and antispasmodic effects, which contribute to the plant?s medicinal value [180, 181]. The anti-inflammatory properties of terpenoids help reduce inflammation, while their antimicrobial action aids in combating infections caused by various microorganisms. Additionally, the antispasmodic activity of these compounds helps in alleviating muscle spasms and related discomforts. Due to these pharmacological effects, M. minuta has been traditionally utilized in treating ailments associated with the respiratory and gastrointestinal systems, where its terpenoid content plays a crucial therapeutic role. The plant's ability to address conditions such as bronchitis, cough, and digestive issues can be linked to these bioactive terpenoids, making M. minuta a valuable resource in herbal medicine [182, 183]. Thus, the presence of terpenoids enhances its application in managing a range of health disorders.
Marsilea minuta, a widely recognized aquatic fern, is an important dietary and medicinal resource due to its rich nutritional composition. Recent investigations have highlighted its significant concentrations of macronutrients and essential biomolecules. Proteins, which play a crucial role in cellular repair and metabolic functions, are present in the plant at levels ranging from 15% to 20% of its dry weight [184]. This protein content positions M. minuta as a valuable plant-based protein source, especially for regions with limited access to animal-derived proteins.?
Carbohydrates, the primary energy-providing biomolecules, are abundantly found in M. minuta, constituting approximately 45% to 50% of its dry weight. This high carbohydrate content underscores the plant's potential as an energy-dense food resource, which could be particularly advantageous in addressing caloric deficiencies in undernourished populations [185]. Additionally, M. minuta contains 12% to 15% dietary fiber by dry weight. The dietary fiber contributes to gastrointestinal health by improving bowel movements, reducing cholesterol levels, and maintaining blood sugar stability, thus rendering M. minuta beneficial for individuals with metabolic disorders.?
Beyond its macronutrient profile, M. minuta also contains notable quantities of essential fatty acids, particularly ?-linolenic acid, a precursor of omega-3 fatty acids. Essential fatty acids are crucial for maintaining cell membrane integrity, regulating inflammatory responses, and supporting cardiovascular and neurological health. The presence of ?-linolenic acid in M. minuta enhances its nutritional value and potential as a functional food.?
Overall, the unique nutrient composition of M. minuta, including its balanced profile of proteins, carbohydrates, dietary fiber, and essential fatty acids, highlights its multifaceted benefits. Its consumption could address various nutritional deficiencies while promoting overall health and well-being, making it an attractive candidate for both dietary supplementation and therapeutic applications. Further research into its bioactive compounds and mechanisms of action could broaden its utility in food science and medicinal contexts.
Table No. 1. Nutritional and Phytochemical Composition of Marsilea minuta
|
Metabolite Category |
Arshin Solomon*, Pragya Pandey, Meghna Singh, Faith Ruth Dixon, Arnab Roy, Akash Bhattacharjee, Unveiling the Medicinal Potential of Dwarf Water Clover (Marsilea minuta): A Comprehensive Review of its Morphological, Anatomical, Phytochemical and Pharmacological Aspects, Int. J. Sci. R. Tech., 2024, 1 (12), 143-178. https://doi.org/10.5281/zenodo.14436029 |